Quantum Mechanics: Fundamental Theory of the Microworld and Modern Technologiesλ
Explore the principles of quantum mechanics, mathematical framework, and practical applications in electronics, photonics, and quantum technologies based on academic sources
Overview
Quantum mechanics describes the behavior of matter at the atomic level — where classical physics breaks down. Wave functions, superposition, the probabilistic nature of measurements 🧬: this isn't philosophy, but a mathematical framework that predicts experimental results to decimal precision. Without quantum mechanics, there would be no transistors, lasers, MRI machines, or quantum computers — 21st-century technology is built on effects that seemed paradoxical a century ago.
🛡️
Laplace Protocol: Quantum mechanics requires rigorous mathematical approaches and experimental verification. Popular speculations about "quantum consciousness" or "remote mental influence" lack scientific foundation and contradict established principles of quantum physics.
Reference Protocol
Scientific Foundation
Evidence-based framework for critical analysis
Protocol: Evaluation
Test Yourself
Quizzes on this topic coming soon
⚡
Deep Dive
Wave Nature of Matter: How Mathematics Describes the Quantum World
Quantum mechanics describes the behavior of matter at atomic and subatomic scales through wave functions and operators. The state of a quantum system is completely characterized by the wave function ψ(r,t), which contains all available information about the system.
The physical meaning of the wave function is revealed through probabilistic interpretation: the square of its modulus |ψ|² determines the probability density of finding a particle at a given point in space. This fundamentally distinguishes the quantum world from classical mechanics, where state is determined by precise coordinates and momenta.
The wave function is not a description of a real wave in space, but a mathematical tool for predicting measurement probabilities. Its modulus squared is the only thing with direct physical meaning.
Schrödinger Equation and Energy Levels
The temporal evolution of a quantum system is described by the Schrödinger equation. It exists in two forms: time-dependent for time-varying processes and time-independent for systems with definite energy.
The time-independent Schrödinger equation Ĥψ = Eψ is an eigenvalue problem. The Hamiltonian operator Ĥ acts on the wave function, yielding discrete energy levels and their corresponding wave functions. This forms the foundation of quantum theory for atoms and molecules.
- Conditions on the Wave Function
- Normalization ∫|ψ|²dV = 1 guarantees that the probability of finding the particle somewhere in space equals unity. Continuity and single-valuedness of ψ impose physical constraints on possible quantum states.
- Superposition Principle
- Linear combinations of solutions to the Schrödinger equation are also solutions. This leads to phenomena of quantum interference and entanglement—effects with no analogs in classical physics.
Operators and Measurable Quantities
Each physical observable corresponds to a linear Hermitian operator acting in the Hilbert space of wave functions. Position is a multiplication operator r̂ = r, momentum is a differentiation operator p̂ = −iℏ∇, energy is the Hamiltonian operator Ĥ = p̂²/2m + V(r̂).
Eigenvalues of operators correspond to possible measurement outcomes, while eigenfunctions represent states with definite values of the given observable. The expectation value of an observable in state ψ is calculated as ⟨Â⟩ = ∫ψ*Âψ dV, connecting mathematical formalism with experimentally measurable quantities.
| Operator | Physical Quantity | Key Property |
|---|---|---|
| r̂ = r | Position | Multiplication by coordinate |
| p̂ = −iℏ∇ | Momentum | Differentiation |
| Ĥ = p̂²/2m + V(r̂) | Energy | Sum of kinetic and potential energy |
Commutation relations between operators determine fundamental limitations on simultaneous measurability of physical quantities. The relation [x̂,p̂ₓ] = iℏ means that position and momentum cannot be simultaneously precisely determined.
Non-commutativity of operators is not a mathematical artifact, but a reflection of the deep asymmetry of quantum reality. Hermiticity of operators guarantees reality of measured values and orthogonality of eigenfunctions.
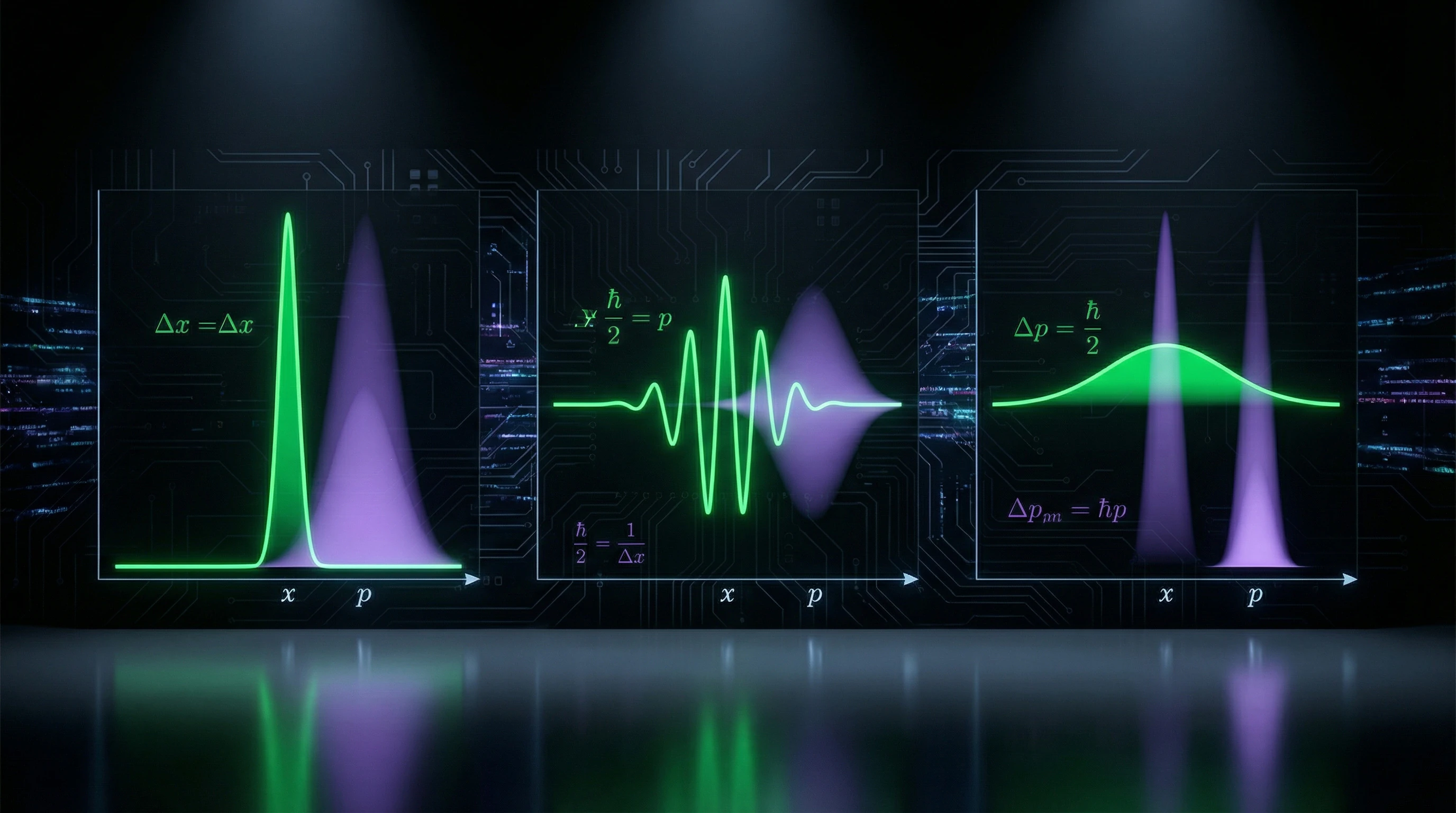
Heisenberg Uncertainty Principle
The uncertainty relation ΔxΔp ≥ ℏ/2 establishes a fundamental limit on the precision of simultaneous measurement of position and momentum. This follows from the non-commutativity of the corresponding operators and reflects the wave nature of matter.
This is not a technical limitation of measuring instruments, but a fundamental property of quantum systems. Analogous relations exist for energy and time ΔEΔt ≥ ℏ/2, which has important consequences for non-stationary processes and virtual states.
- The more precisely position is determined (small Δx), the greater the uncertainty in momentum (large Δp)
- The more precisely energy is determined (small ΔE), the greater the uncertainty in the lifetime of the state (large Δt)
- These limitations are universal and independent of the specific system or measurement method
Solutions to Model Problems: From Potential Wells to the Hydrogen Atom
Exact analytical solutions to the Schrödinger equation exist only for a limited number of model systems, which play a key role in understanding quantum phenomena and serve as the foundation for approximation methods. These standard problems—particle in a potential well, harmonic oscillator, and hydrogen atom—demonstrate fundamental quantum effects: discrete energy spectra, tunneling, and quantization of angular momentum.
Mathematical methods developed for these systems are applied in solid-state physics, quantum optics, and semiconductor device theory.
Particle in a Potential Well
The infinite potential well—the simplest quantum system where a particle is confined by impenetrable walls in the region 0 < x < L. Solution of the stationary Schrödinger equation yields a discrete spectrum Eₙ = n²π²ℏ²/2mL² and wave functions ψₙ(x) = √(2/L)sin(nπx/L), where n = 1,2,3...
The ground state energy E₁ = π²ℏ²/2mL² is nonzero—a fundamental difference from classical mechanics and a consequence of the uncertainty principle.
| Parameter | Infinite Well | Finite Well |
|---|---|---|
| Spectrum | Discrete, n² | Discrete, limited number of levels |
| Wave function beyond boundary | Zero | Exponential decay ψ ~ exp(−κx) |
| Application | Qualitative understanding | Quantum wells in semiconductors |
A finite potential well of depth V₀ allows penetration of the wave function into the classically forbidden region. The number of bound states is determined by the parameter V₀L²m/ℏ².
The tunneling effect—passage of a particle through a potential barrier with energy less than its height—is described by the transmission coefficient T ~ exp(−2κd), where d is the barrier width. This effect underlies tunnel diodes and scanning tunneling microscopes.
Quantum Harmonic Oscillator
The harmonic oscillator with potential V(x) = mω²x²/2 is one of the most important models in quantum mechanics, applicable to atomic vibrations in molecules, phonons in crystals, and quantization of the electromagnetic field. The energy spectrum Eₙ = ℏω(n + 1/2) is equidistant with spacing ℏω.
The zero-point energy E₀ = ℏω/2 reflects the quantum nature of the system: even in the ground state, the oscillator cannot be at rest.
Wave functions are expressed through Hermite polynomials Hₙ(ξ) and a Gaussian function: ψₙ(x) ~ Hₙ(x√(mω/ℏ))exp(−mωx²/2ℏ).
The operator method using creation â⁺ and annihilation â operators allows solving the problem without explicit integration. These operators satisfy the commutation relation [â,â⁺] = 1 and act as â⁺|n⟩ = √(n+1)|n+1⟩ and â|n⟩ = √n|n−1⟩.
- From the ground state |0⟩, the entire ladder of energy levels is constructed
- Each action of the creation operator raises the system by one level
- The annihilation operator lowers the system until it reaches the ground state
- This formalism extends to quantized fields in quantum electrodynamics
Hydrogen Atom and Hydrogen-Like Systems
The hydrogen atom with Coulomb potential V(r) = −e²/4πε₀r is the only real system for which the Schrödinger equation is solved exactly in the three-dimensional case. The energy spectrum Eₙ = −13.6 eV/n² is determined by the principal quantum number n = 1,2,3...
Wave functions are characterized by three quantum numbers: n (energy), l (orbital angular momentum), and m (momentum projection). The radial part is expressed through Laguerre polynomials, the angular part through spherical harmonics Yₗᵐ(θ,φ), reflecting the spherical symmetry of the problem.
- Angular Momentum Quantization
- L² = ℏ²l(l+1), where l = 0,1,...,n−1. Leads to the shell structure of the atom and explains the periodic table of elements.
- Momentum Projection
- Lz = ℏm, where m = −l,...,+l. Discreteness of projection is a purely quantum effect absent in classical physics.
- Hydrogen-Like Ions
- He⁺, Li²⁺ are described by the same structure with substitution e² → Ze². Allows calculation of spectra and is applied in plasma spectroscopy.
Fine structure of spectral lines, caused by spin-orbit interaction and relativistic corrections, requires accounting for electron spin and the Dirac equation. These effects lead to splitting of energy levels and explain observed spectra with high precision.
Perturbation Theory Methods: Calculating Quantum Transitions and Spectra
Perturbation theory is a systematic method for approximate solution of quantum mechanical problems when the Hamiltonian can be represented as Ĥ = Ĥ₀ + λV̂. Here Ĥ₀ is an exactly solvable problem, and λV̂ is a small perturbation.
Corrections to energies and wave functions are calculated as power series in λ. The method applies to atoms in electric fields, interaction of radiation with matter, and other systems. We distinguish between time-independent theory (constant perturbation) and time-dependent theory (time-varying perturbation).
Time-Independent Perturbation Theory
First-order correction to the energy of the nth level: E⁽¹⁾ₙ = ⟨ψ⁽⁰⁾ₙ|V̂|ψ⁽⁰⁾ₙ⟩, where ψ⁽⁰⁾ₙ is the unperturbed wave function.
Second-order correction E⁽²⁾ₙ = Σₖ≠ₙ |⟨ψ⁽⁰⁾ₖ|V̂|ψ⁽⁰⁾ₙ⟩|²/(E⁽⁰⁾ₙ - E⁽⁰⁾ₖ) accounts for virtual transitions to intermediate states. It determines atomic polarizability and dispersion forces.
Applicability condition: matrix elements of the perturbation must be small compared to differences in unperturbed energies: |⟨k|V̂|n⟩| ≪ |E⁽⁰⁾ₙ - E⁽⁰⁾ₖ|.
When the unperturbed level is degenerate, degenerate perturbation theory is applied: first diagonalize the perturbation matrix in the subspace of degenerate states.
| Effect | Field | Splitting Dependence |
|---|---|---|
| Stark | Electric | Quadratic (hydrogen ground state) |
| Zeeman | Magnetic | Linear, proportional to m |
Time-Dependent Perturbation Theory and Selection Rules
Time-dependent theory describes transitions under time-dependent perturbation V̂(t). Applicable to interaction of atoms with electromagnetic radiation.
Transition amplitude from state |i⟩ to |f⟩ in first order: cₓ(t) = -(i/ℏ)∫₀ᵗ⟨f|V̂(t')|i⟩exp(iωₓᵢt')dt', where ωₓᵢ = (Eₓ - Eᵢ)/ℏ. For harmonic perturbation V̂(t) = V̂cos(ωt), transition probability is maximal at resonance ω ≈ ωₓᵢ.
The resonance condition explains selective light absorption by atoms: the system responds only to frequencies matching differences in its energy levels.
Selection rules determine allowed and forbidden transitions. For electric dipole transitions in the hydrogen atom: Δl = ±1 and Δm = 0,±1. These rules follow from properties of matrix elements of the dipole moment operator d̂ = -er̂.
Forbidden transitions have zero matrix elements in the dipole approximation, but can occur through quadrupole or magnetic dipole mechanisms with substantially lower probabilities.
Transition Probabilities and Lifetimes
Fermi's golden rule determines the transition probability per unit time for interaction with a continuous spectrum of final states: wᵢ→ₓ = (2π/ℏ)|⟨f|V̂|i⟩|²ρ(Eₓ), where ρ(Eₓ) is the density of final states.
Applicable to calculating rates of radiative transitions, photoelectric effect, and particle scattering. Lifetime of an excited state τ = 1/Σₓwᵢ→ₓ is determined by the sum of probabilities of all possible transitions to lower-lying states.
- Natural linewidth
- Γ = ℏ/τ — related to the state lifetime. Shorter lifetime means broader line.
- Einstein A coefficient (spontaneous emission)
- Aₓᵢ = (ω³ₓᵢ/3πε₀ℏc³)|dₓᵢ|², where dₓᵢ is the dipole moment matrix element.
- Ratio of spontaneous to stimulated emission
- Aₓᵢ/Bₓᵢu(ω) = ℏω³/π²c³ increases with frequency. Explains dominance of spontaneous emission in the optical range and the necessity of population inversion for lasers.
Quantum Phenomena and Effects in Microworld and Macrostructures
Tunneling Effect and Barrier Penetration
The tunneling effect is a quantum mechanical phenomenon in which a particle penetrates through a potential barrier whose height exceeds its kinetic energy. In classical mechanics, this is impossible.
The barrier transmission coefficient D ≈ exp(−2κa), where κ = √(2m(U₀−E)/ℏ²). The tunneling probability decreases exponentially with increasing particle mass and barrier width—which is why the effect is observed predominantly for electrons and light particles.
The tunneling effect underlies alpha decay of nuclei, field emission, and the operation of tunnel diodes. In semiconductor heterostructures, resonant tunneling diodes achieve switching speeds up to terahertz frequencies.
The reflection coefficient R = 1 − D demonstrates oscillating behavior as particle energy changes. Resonant tunneling occurs when the energy coincides with quasi-stationary levels in the potential well.
Angular Momentum and Particle Spin
Angular momentum in quantum mechanics is quantized: L² = ℏ²l(l+1), where l = 0, 1, 2, ..., and the projection Lz = ℏm, where m = −l, −l+1, ..., l. For each l there exist 2l+1 distinct states, which determines the degeneracy of energy levels in centrally symmetric potentials.
Orbital angular momentum is associated with spatial motion and is described by spherical harmonics Yₗₘ(θ,φ), which determine the angular dependence of the wave function.
| Characteristic | Orbital Angular Momentum | Spin |
|---|---|---|
| Source | Spatial motion | Intrinsic angular momentum |
| For electron | l = 0, 1, 2, ... | s = 1/2 |
| Projections | 2l+1 values | 2 values (±ℏ/2) |
Total angular momentum J is combined according to quantum addition rules: J² = ℏ²j(j+1), where j = |l−s|, ..., l+s. Spin-orbit interaction leads to fine structure of atomic spectra: ΔE ~ α²mc²(Z/n)⁴, where α ≈ 1/137 is the fine structure constant.
Quantum Statistics and the Pauli Principle
The indistinguishability of quantum particles creates a fundamental difference in statistics: fermions (half-integer spin) obey Fermi-Dirac statistics, bosons (integer spin) obey Bose-Einstein statistics.
The Pauli exclusion principle forbids two fermions from occupying the same quantum state. The wave function is antisymmetric: ψ(r₁,r₂) = −ψ(r₂,r₁). For bosons, the wave function is symmetric, allowing an unlimited number of particles in the same state.
The Fermi-Dirac distribution function f(E) = 1/(exp[(E−μ)/kT]+1) determines the probability of occupying a state with energy E at temperature T and chemical potential μ.
- As T → 0, all states with E < μ are filled, those with E > μ are empty
- The Fermi energy EF and Fermi momentum pF = ℏkF characterize the degenerate electron gas
- Density of states g(E) and distribution function determine thermodynamic properties
- Heat capacity C ~ T at low temperatures for fermions (deviation from the classical Dulong-Petit law)

Applications in Electronics and Photonics of Modern Devices
Semiconductor Physics and Quantum Wells
In semiconductor heterostructures, band discontinuities at material interfaces create potential wells for charge carriers. Quantized energy levels are determined by the formula En = ℏ²π²n²/(2m*L²), where m* is the effective mass, n = 1, 2, 3, .... When well thickness reaches nanometers, the spacing between levels becomes comparable to thermal energy kT or photon energy.
Density of states in quantum wells is stepwise: g₂D(E) = m*/(πℏ²) for each subband, unlike the parabolic g₃D(E) ~ √E in bulk. This sharply increases the density of states near subband edges and improves laser characteristics: reduces threshold current, increases gain, and enhances temperature stability.
| Parameter | Bulk Semiconductor | Quantum Well |
|---|---|---|
| Density of States | g₃D(E) ~ √E (parabolic) | g₂D(E) = const (stepwise) |
| Laser Threshold Current | Higher | Lower |
| Temperature Stability | Worse | Better |
Quantum Electronics and Lasers
Laser generation requires population inversion: N₂ > N₁. The gain coefficient g = σ(N₂−N₁) must exceed cavity losses α. The threshold current of an injection laser Ith = eVd(N₂−N₁)th/τ determines the minimum pump power.
Quantum cascade lasers utilize intersubband transitions in coupled quantum well systems. An electron sequentially emits photons while passing through multiple active regions, and each injected electron generates several photons — quantum efficiency exceeds 100%.
Emission wavelength λ = hc/ΔE is determined by the subband energy difference and can be tuned by changing well thickness. This allows coverage of a wide mid- to far-infrared spectral range with a single device architecture.
Fiber-Optic Communication Systems
Light propagation in optical fiber is described by the wave equation with refractive index profile n(r). For step-index fiber, the normalized frequency V = (2πa/λ)NA determines the number of guided modes: single-mode operation requires V < 2.405.
At λ = 1.55 μm, this corresponds to a core diameter of approximately 9 μm. Modal group velocity dispersion causes pulse broadening Δτ ≈ (n₁Δn/c)L, which limits transmission speed in multimode fibers.
Nonlinear effects at high intensities: Kerr nonlinearity causes self-phase modulation, stimulated Raman scattering transfers energy to the Stokes component. Solitons — pulses that maintain their shape through a balance of dispersion and nonlinearity — are described by the nonlinear Schrödinger equation and are used for ultra-long-distance transmission without regeneration.
Solitons demonstrate a fundamental principle: opposing effects (dispersion and nonlinearity) in precise balance create a stable structure capable of traveling thousands of kilometers without distortion.
Modern Directions in Quantum Technologies and Development Prospects
Quantum Dots and Nanostructures
Quantum dots are semiconductor nanostructures with dimensional quantization in all three dimensions, yielding a completely discrete energy spectrum similar to an atom. The ground state energy E₀ = ℏ²π²/(2m*)(1/Lx² + 1/Ly² + 1/Lz²) depends on geometry, allowing optical properties to be controlled by changing size.
The density of states g₀D(E) = Σδ(E-En) represents a set of delta functions, providing maximally narrow spectral lines and high efficiency of radiative transitions.
Self-assembled quantum dots form during epitaxial growth of strained heterostructures via the Stranski-Krastanov mechanism: after depositing a critical thickness of the wetting layer, spontaneous formation of three-dimensional islands occurs to relax elastic strain.
Typical dimensions of InAs/GaAs quantum dots are 10–30 nm at the base and 3–8 nm in height, corresponding to dimensional quantization energies of 50–200 meV and emission wavelengths of 1.0–1.3 μm.
Quantum dot lasers demonstrate record-low threshold currents (less than 10 A/cm²), weak temperature dependence, and narrow spectral linewidth of generation.
Supersymmetric Quantum Mechanics
Supersymmetric quantum mechanics (SUSYQM) connects pairs of Hamiltonians H₁ and H₂ through supersymmetric operators Q and Q†: H₁ = Q†Q, H₂ = QQ†. The spectra of partner Hamiltonians are isospectral, except for a possible zero-level ground state.
The superpotential W(x) determines the form of partner potentials: V₁(x) = W²(x) - ℏW'(x)/√(2m), V₂(x) = W²(x) + ℏW'(x)/√(2m).
- The factorization method allows construction of hierarchies of isospectral potentials with successively removed lower energy levels.
- Application in inverse problems of quantum mechanics and construction of potentials with specified scattering properties.
- Supersymmetry breaking occurs when the ground state of one of the partner Hamiltonians has zero energy, analogous to spontaneous symmetry breaking in field theory.
Biorthogonal Quantum Mechanics
Biorthogonal quantum mechanics generalizes the standard formalism to the case of non-Hermitian Hamiltonians, using different bases for bra- and ket-vectors: ⟨φn|ψm⟩ = δnm, where {|ψn⟩} and {⟨φn|} are right and left eigenvectors.
PT-symmetric Hamiltonians, invariant under the combined operation of spatial inversion P and time reversal T, can have a completely real spectrum despite non-Hermiticity. The PT-symmetry condition H(x,p) = H(-x,-p)* leads to specific properties of wave functions and selection rules for transitions.
The biorthogonal formalism finds application in describing open quantum systems with dissipation, resonant states, and quasi-stationary levels with finite lifetime.
The metric operator η connects the biorthogonal basis with the orthonormal one through a similarity transformation, defining the physical norm of states: ⟨ψ|η|ψ⟩ must be positive definite.
PT-symmetry breaking at exceptional points in the spectrum leads to coalescence of eigenvalues and eigenvectors, which is used to create ultra-sensitive sensors and optical signal amplifiers.

Knowledge Access Protocol
FAQ
Frequently Asked Questions
Quantum mechanics is a branch of physics that describes the behavior of particles at the atomic and subatomic level. Unlike classical physics, particles here exhibit wave properties, and their state is described by probabilities. This theory underlies modern electronics, lasers, and semiconductor technologies.
A wave function describes the probability of finding a particle at a specific point in space, rather than an exact trajectory. The square of the wave function's magnitude gives the probability density of finding the particle. This is a fundamental difference from classical mechanics, where the trajectory is uniquely determined.
The principle states that it is impossible to simultaneously measure a particle's position and momentum with perfect accuracy. The more precisely we measure position, the less defined the momentum becomes, and vice versa. This is not a limitation of instruments, but a fundamental property of the quantum world.
No, this is a common myth. Quantum entanglement does not allow information to be transmitted faster than light or objects to be influenced mentally. Real quantum effects are constrained by strict physical laws and have no connection to paranormal phenomena.
Yes, many familiar technologies are based on quantum effects. LEDs, lasers, transistors in smartphones, solar panels—all of these work thanks to quantum mechanics. Quantum phenomena also manifest in superconductivity and the operation of MRI scanners.
The tunnel effect is the passage of a particle through a potential barrier that it could not classically overcome. This phenomenon is used in tunnel diodes, scanning tunneling microscopes, and underlies nuclear fusion in stars. The probability of tunneling depends exponentially on the height and width of the barrier.
Begin by mastering the mathematical apparatus: linear algebra, differential equations, and operator theory. Then study basic concepts—the wave function, Schrödinger's equation, simple systems (potential well, oscillator). Solve problems in parallel to develop physical intuition.
For most real systems, approximation methods are used, especially perturbation theory. A complex system is represented as a simple one with small additions, for which corrections to energy and wave functions are found. Numerical methods and the variational principle are also applied.
Perturbation theory allows finding approximate solutions for systems that cannot be solved exactly. The method separates the problem into a solvable part and a small correction, yielding practical results for atoms, molecules, and solids. It is the primary tool for calculating quantum systems.
A quantum well is a thin semiconductor layer between layers with a larger bandgap, where electrons are confined in one dimension. Such structures are used in laser diodes, high-brightness LEDs, and high-speed transistors. Energy quantization in the well determines optical and electrical properties.
Spin is an intrinsic quantum angular momentum of a particle, unrelated to motion through space. The electron has spin 1/2, which has no classical analogue. Spin determines the magnetic properties of particles and their statistics (fermions or bosons).
Yes, but not because of 'observer consciousness.' Measurement requires physical interaction between the apparatus and the system, which inevitably disturbs its state and causes wavefunction collapse. This is an objective physical process, independent of human presence.
Lasers are based on stimulated emission—a quantum transition of atoms from an excited state under the influence of a photon. This creates coherent light of a single frequency and phase. Population inversion and a resonator provide amplification of the radiation.
This is an extension of quantum mechanics that connects bosonic and fermionic states through supersymmetry. The method allows finding exact solutions for certain potentials and constructing hierarchies of isospectral Hamiltonians. It is applied in theoretical physics and mathematical physics to study integrable systems.
In principle yes, but quantum effects are rapidly suppressed by decoherence through interaction with the environment. Exceptions include superconductors, superfluids, and Bose-Einstein condensates, where macroscopic quantum states are stable. Quantum computers aim to exploit quantum properties of mesoscopic systems.
This is a generalization of standard quantum mechanics for non-Hermitian Hamiltonians using two sets of states—'bra' and 'ket.' The approach is applied to describe open quantum systems, resonances, and systems with amplification or decay. It enables working with PT-symmetric systems.
