Physics: The Fundamental Science of Matter, Energy, and Their Interactionsλ
Exploring physical phenomena through theoretical knowledge and experimental practice, from mechanics and molecular physics to quantum theory and atomic physics
Overview
Physics isn't a collection of formulas to memorize—it's a tool 🧩 for decoding reality: from the trajectory of a thrown stone to the quantum states of an atom. Effective learning requires linking theory with experiment—measure, make mistakes, analyze uncertainties, understand why a law works, not just how to apply it. Programs are built hierarchically (mechanics → thermodynamics → quantum physics), but the goal is singular: learn to see physical principles in real problems—from engineering calculations to technological breakthroughs.
🛡️
Laplace Protocol: Experimental physics and methodological knowledge are not secondary but fundamental components of physics education. Understanding measurement uncertainty is not an admission of error, but a key concept of the scientific method.
Reference Protocol
Scientific Foundation
Evidence-based framework for critical analysis
Protocol: Evaluation
Test Yourself
Quizzes on this topic coming soon
⚡
Deep Dive
Methodological Foundations of Physics: How Science Studies Nature
Methods of Studying Physical Phenomena
Physics relies on systematic study of matter, energy and their interactions through a combination of theoretical and experimental approaches. Methodological knowledge — understanding scientific methods, experimental approaches and research techniques — is fundamental to grasping physical content.
An integrated approach combining theory with experimental practice is recognized by the scientific community as a necessary condition for quality physics education. Experimental physics is the practical application of physical principles through observation, measurement and experimentation to verify theoretical predictions.
Common misconception: physics can be studied purely theoretically. In reality, experimental work is necessary for understanding physical phenomena and developing scientific thinking.
Progressive educational programs systematically incorporate laboratory work, starting with basic measurements and progressing to complex research projects.
Measurements and Uncertainties as the Foundation of Experimental Physics
Understanding measurement methods and error analysis forms the foundation of physics education. Measurement uncertainty is not a mistake to be avoided, but a fundamental concept of experimental physics, reflecting the inevitable uncertainty of any measurement.
- Physical Quantities
- Classified by criteria: scalar and vector, fundamental and derived, extensive and intensive. The International System of Units (SI) ensures standardization through seven base units: meter, kilogram, second, ampere, kelvin, mole and candela.
- Correct Use of Units
- Prevents conceptual errors and ensures reproducibility of results. Systematic training in measurement methods should begin at early stages and deepen as complex experimental techniques are mastered.
The Scientific Method in Physics and Its Application
The scientific method in physics is a cyclical process: observation → hypothesis → experimental verification → data analysis → conclusions. Methodological understanding is fundamental to grasping physical content and conducting meaningful research.
| Component of Scientific Method | Role in Physics |
|---|---|
| Observation and Hypothesis | Formulating a question that can be tested experimentally |
| Experimental Verification | Collecting data under controlled conditions |
| Analysis and Critical Evaluation | Interpreting results and identifying limitations |
| Transformation into Engineering Solutions | Applying physical principles in technologies |
Modern educational programs emphasize development of research skills through independent projects and laboratory work. Students must master not only theoretical concepts, but also practical skills: experiment planning, data collection, statistical analysis and critical evaluation of results.
Historical context, including contributions from international physicists, demonstrates the evolution of the scientific method. The connection between physical principles and technological applications requires understanding how the scientific method transforms into engineering solutions.
Mechanics and Fundamentals of Motion: From Kinematics to Conservation Laws
Kinematics and Dynamics of a Material Point
Mechanics studies the motion of bodies and interactions between them, forming the foundation for understanding more complex physical phenomena. Kinematics describes motion without considering its causes, using concepts of trajectory, displacement, velocity, and acceleration.
Dynamics investigates the causes of motion through Newton's laws, connecting force, mass, and acceleration into a unified conceptual system. Educational programs traditionally begin with mechanics because its concepts are most accessible for direct observation and experimental verification.
- Law of inertia: a body maintains its state of rest or uniform motion without external forces.
- Fundamental law of dynamics: acceleration is proportional to applied force and inversely proportional to mass.
- Law of action-reaction: interaction forces are equal in magnitude and opposite in direction.
The concept of an inertial reference frame is critically important for correct application of Newton's laws and understanding the relativity of motion. Forces of different nature—gravitational, elastic, friction—are studied as specific manifestations of fundamental interactions, each with characteristic mathematical models and experimental research methods.
Laws of Conservation of Energy and Momentum
Conservation laws represent fundamental principles of physics, asserting the invariance of certain quantities in isolated systems. The law of conservation of energy postulates that the total energy of a closed system remains constant, though it may transform between kinetic, potential, and other forms.
Conservation laws possess greater generality than Newton's laws, applying even in relativistic and quantum mechanics.
The law of conservation of momentum is especially important for analyzing collisions and interactions where external forces are absent or negligibly small. Work and power quantitatively describe energy transfer processes when forces act on moving bodies.
Potential energy is associated with system configuration and conservative forces such as gravity and elasticity, while kinetic energy is determined by the mass and velocity of a moving object. Practical applications of conservation laws span a wide spectrum of technologies—from simple mechanisms to spacecraft, demonstrating the connection between fundamental physics and engineering.
Mechanical Oscillations and Waves
Oscillatory processes represent periodic changes in physical quantities relative to an equilibrium state and occur in all branches of physics. Harmonic oscillations are described by sinusoidal functions and characterized by amplitude, frequency, period, and phase.
The mathematical pendulum and spring oscillator serve as classical models for studying oscillatory systems, demonstrating the interconversion of kinetic and potential energy. Damped oscillations account for dissipative forces, while forced oscillations and resonance have critical significance for understanding many technical systems.
- Free oscillations: the system oscillates without external influence after initial disturbance.
- Damped oscillations: amplitude decreases due to friction and medium resistance.
- Forced oscillations: external periodic force maintains or changes oscillations.
- Resonance: maximum amplitude when external force frequency coincides with the system's natural frequency.
Mechanical waves represent the propagation of oscillations in an elastic medium without matter transfer, only with transmission of energy and momentum. Longitudinal waves are characterized by particle oscillations along the direction of propagation, while transverse waves oscillate perpendicular to this direction.
Sound waves as an example of longitudinal mechanical waves are studied from the perspective of their physical characteristics—frequency, wavelength, propagation speed—and their connection to physiological perception. Phenomena of interference, diffraction, and standing waves demonstrate wave nature and find applications in acoustics and engineering.
Molecular Physics and Thermodynamics: From Particle Chaos to Macroscopic Laws
Kinetic Molecular Theory of Matter
Molecular physics connects the microscopic behavior of particles with macroscopic observable quantities. Matter consists of discrete particles in continuous random motion — the intensity of this motion is temperature.
Brownian motion experimentally confirms the molecular structure of matter. Molecular sizes (nanometers) and Avogadro's number link the microscopic and macroscopic scales.
| Concept | Definition | Connection to Observable |
|---|---|---|
| Ideal Gas | Model neglecting molecular size and interactions (except elastic collisions) | Pressure ↔ average kinetic energy |
| Temperature (Kelvin) | Directly proportional to average kinetic energy of molecules | Link between thermodynamics and statistical mechanics |
| Maxwell Distribution | Statistical distribution of molecular velocities at equilibrium | Predicts macroscopic properties of gas |
Fundamentals of Thermodynamics and Its Laws
Thermodynamics studies macroscopic properties of systems and energy transformation without detailed analysis of molecular structure. The first law is the conservation of energy for thermal processes: the change in internal energy equals the sum of heat transferred and work done.
The internal energy of an ideal gas depends only on temperature and is determined by the total kinetic energy of random molecular motion. Heat transfer occurs through three pathways: conduction, convection, radiation — each with its own governing principles.
The second law of thermodynamics establishes the direction of spontaneous processes through entropy — a measure of system disorder. The impossibility of a perpetual motion machine of the second kind reflects the fundamental asymmetry of time.
Heat engines convert internal energy into mechanical work with efficiency limited by the Carnot cycle. Refrigerators and heat pumps use reverse cycles to transfer heat from cold to hot bodies using external work.
Properties of Gases, Liquids, and Solids
The ideal gas law relates pressure, volume, temperature, and amount of substance for dilute gases. Isoprocesses (isothermal, isobaric, isochoric, adiabatic) — idealized processes with one constant parameter, used to analyze real cycles.
Real gases deviate from ideal behavior at high pressures and low temperatures. The Van der Waals equation accounts for corrections due to molecular volume and intermolecular interactions.
- Liquids
- Short-range molecular order; surface tension, capillarity, viscosity — unique properties absent in gases.
- Crystalline Solids
- Long-range order and anisotropy of properties; amorphous solids are isotropic and lack a definite melting temperature.
- Phase Transitions
- Melting, evaporation, sublimation are accompanied by absorption or release of latent heat without temperature change; phase diagrams display regions of phase existence.
Experimental Physics: From Measurement to Discovery of Patterns
Fundamentals of Physical Experimentation and the Scientific Method
Experimental physics is a systematic approach to studying nature through observation, measurement, and verification of theoretical predictions. The scientific method: hypothesis → planning → measurements → data analysis → conclusions.
Methodological knowledge of experimental approaches and research techniques is fundamental to grasping physical content. Physics education requires integration of theory with practice—purely theoretical study without experimental work does not provide complete understanding of phenomena.
Experiment without theory is blind searching. Theory without experiment is empty speculation. Physics lives in their dialogue.
Measurement Instruments and Methods of Physical Measurement
Measurement methods, classification of physical quantities, and error analysis form the foundation of physics education. Physical quantities are divided into fundamental (length, mass, time, electric current, temperature, amount of substance, luminous intensity) and derived quantities, expressed through International System (SI) units.
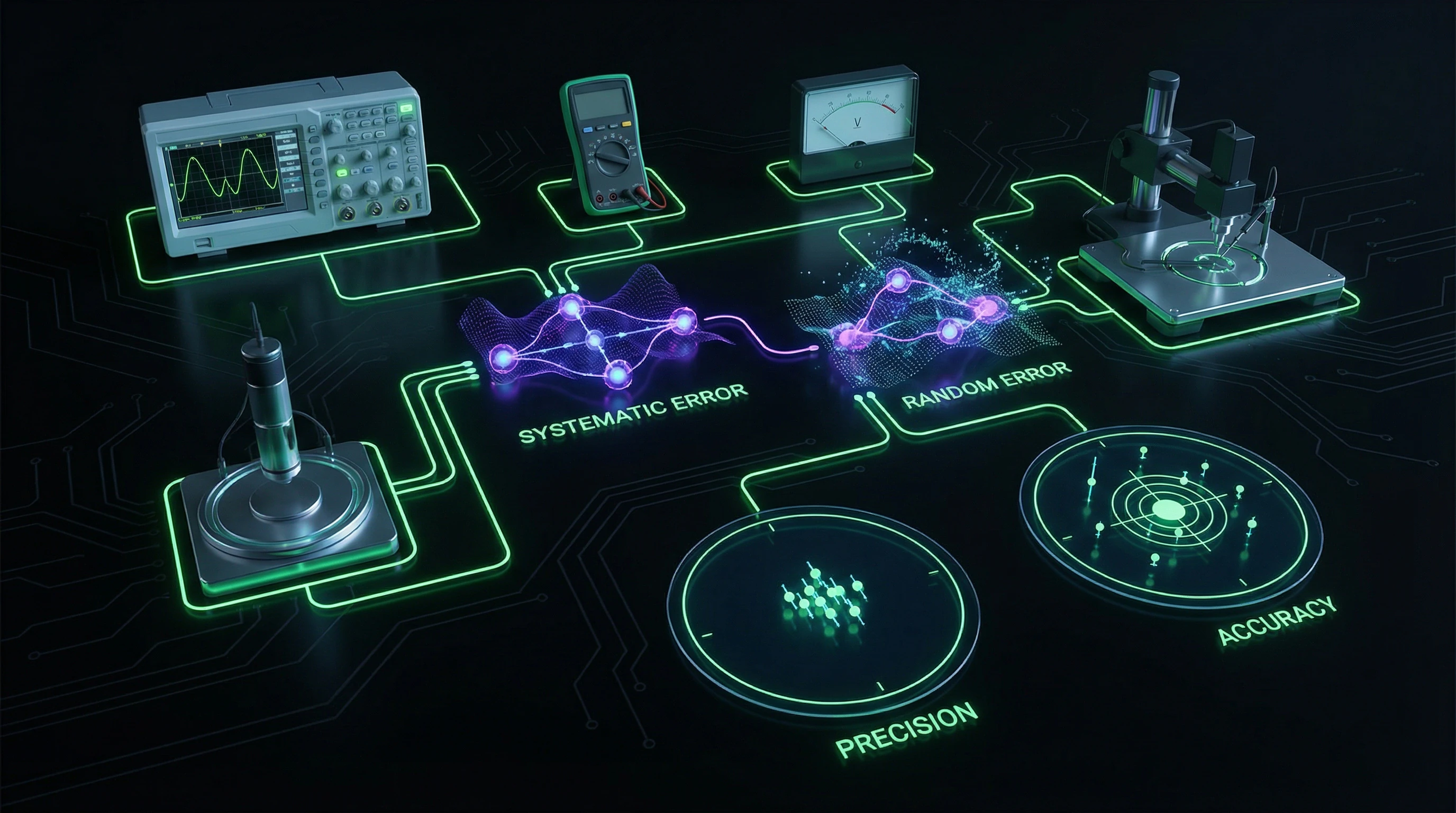
Measurement instruments encompass mechanical tools (rulers, calipers, micrometers), electrical devices (ammeters, voltmeters, ohmmeters), optical instruments, and digital sensors. Every measurement is accompanied by uncertainty—an indeterminacy that is not a mistake but represents a fundamental characteristic of experimental physics.
| Type of Uncertainty | Source | Method of Accounting |
|---|---|---|
| Random | Fluctuations in conditions, scatter in readings | Multiple measurements, standard deviation |
| Systematic | Improper calibration, external factors | Instrument calibration, accounting for external conditions |
Processing Experimental Data and Statistical Analysis
Data processing includes calculating mean values, determining absolute and relative uncertainties, constructing graphs, and identifying functional dependencies. Statistical methods assess random uncertainties through repeated measurements and calculation of standard deviation.
Graphical representation of data—plotting dependencies of physical quantities with confidence intervals—facilitates visualization of patterns and verification of theoretical models. Correct data processing and understanding of uncertainty develop critical thinking and form the scientific literacy necessary for interpreting research results.
Data without uncertainties is not data, but faith. An honest experiment shows the boundaries of its knowledge.
Quantum and Atomic Physics: The Microworld and Its Laws
Quantum Phenomena and Wave-Particle Duality
At atomic and subatomic scales, classical physics laws cease to work—quantum mechanics takes over. Microparticles (electrons, photons) exhibit both wave and particle properties simultaneously, depending on observation conditions.
The photoelectric effect demonstrates the particle nature of light: electrons are ejected from metal only when a threshold frequency is exceeded. Photon energy E = hν, where h is Planck's constant and ν is frequency.
- de Broglie Wavelength
- λ = h/p, where p is particle momentum. Electron diffraction and interference confirm their wave nature and show that duality is not a metaphor but a fundamental property of the microworld.
Atomic Structure and Quantum States
The atomic nucleus (~10⁻¹⁵ m) is five orders of magnitude smaller than the atom itself (~10⁻¹⁰ m), yet contains nearly all its mass. Electrons occupy space around the nucleus according to quantum rules, not classical orbits.
The Bohr model postulates stationary energy levels: electrons move without radiation, and transitions between levels are accompanied by emission or absorption of photons with energy ΔE = hν.
| Quantum Number | Symbol | What It Determines |
|---|---|---|
| Principal | n | Electron energy and orbital size |
| Orbital | l | Shape of electron cloud |
| Magnetic | m | Orbital orientation in space |
| Spin | s | Intrinsic angular momentum of electron |
Atomic spectral lines are unique "fingerprints" of elements. They arise from quantum transitions and are used in spectral analysis for substance identification.
Nuclear Physics and Radioactivity
The nucleus consists of protons and neutrons held together by the strong nuclear force, which overcomes electrostatic repulsion between protons. This interaction operates only at distances of ~10⁻¹⁵ m.
Radioactivity is the spontaneous transformation of unstable nuclei. Each isotope has its own half-life, making radioactivity predictable in a statistical sense but unpredictable for individual nuclei.
Nuclear reactions—fission of heavy nuclei and fusion of light nuclei—release enormous energy according to E = mc². This energy underlies nuclear power and thermonuclear fusion.
- α-radiation: helium nuclei (2 protons + 2 neutrons)
- β-radiation: electrons or positrons from the nucleus
- γ-radiation: high-energy photons
Dosimetry and protection from ionizing radiation are critically important for safe use of radioactive materials in medicine, industry, and scientific research.

Physics as the Foundation of Technology: From Theory to Practice
Application of Physical Principles in Modern Technology
Physics serves as the foundation for technology and engineering. Mechanics underlies the design of machines, vehicles, and structures — Newton's laws and conservation principles determine the strength, stability, and dynamics of systems.
Thermodynamics enables the operation of heat engines, refrigeration systems, and power plants, where energy conversion efficiency is limited by the second law. Electrodynamics and optics form the basis of electrical engineering, telecommunications, and laser technologies.
| Field of Physics | Technological Applications |
|---|---|
| Mechanics | Machinery, transportation, construction |
| Thermodynamics | Heat engines, power generation, air conditioning |
| Electrodynamics | Electrical engineering, telecommunications, fiber optics |
| Quantum Physics | Semiconductors, computers, nanotechnology |
Historical Context and Contributions of Distinguished Physicists
Isaac Newton laid the foundations of classical mechanics and gravitation. James Maxwell created the theory of electromagnetism, Albert Einstein developed the theory of relativity and the quantum theory of the photoelectric effect.
Russian scientists made fundamental contributions: Mikhail Lomonosov formulated the law of conservation of matter and motion, Dmitri Mendeleev created the periodic table of elements, Alexander Popov invented radio, and Pyotr Kapitsa investigated low-temperature physics and superfluidity.
Understanding the historical development of physical concepts helps illuminate the evolution of scientific thinking and the connection between theoretical discoveries and technological progress.
Contemporary Directions in Physics Research
Modern physics advances in the fields of particle physics, cosmology, condensed matter physics, and nanophysics. High-energy physics investigates fundamental particles at accelerators like the Large Hadron Collider, testing the Standard Model.
Quantum technologies — quantum computers, quantum cryptography, quantum sensors — promise revolutionary breakthroughs in computation, information security, and measurement precision. Condensed matter physics studies superconductivity, graphene, and topological materials with unique properties.
- Quantum computers — revolution in computational power
- Quantum cryptography — absolute information security
- Quantum sensors — precision measurements
- Superconductivity — lossless energy technologies
- Metamaterials — materials with engineered properties
Interdisciplinary research at the intersection of physics, chemistry, biology, and computer science is forming new fields of knowledge and technological platforms for the 21st century.
Knowledge Access Protocol
FAQ
Frequently Asked Questions
Physics is a natural science that studies matter, energy, and their interactions. It investigates the fundamental laws of nature, from the motion of objects to quantum phenomena, and serves as the foundation for understanding physical processes and developing technologies. Physics encompasses mechanics, thermodynamics, electrodynamics, optics, quantum physics, and nuclear physics.
Experimental work is necessary for understanding physical phenomena and developing scientific thinking. Without hands-on practice, it's impossible to master measurement methods, learn to analyze data, and understand the real behavior of physical systems. Experimentation connects theory with reality and develops research skills.
Measurement uncertainty is the inevitable deviation of a result from the true value of a quantity. Understanding uncertainty is a fundamental concept in experimental physics, not a sign of error. Proper assessment of uncertainties allows you to determine the reliability of results and the boundaries of applicability of conclusions.
Begin by mastering methods for measuring physical quantities and units of measurement. Then systematically study mechanics, molecular physics, thermodynamics, and continue through the curriculum. Always combine theory with problem-solving and performing experiments to reinforce the material.
No, this is a common misconception. Methodological understanding is the foundation for meaningful study of physics and conducting research. Knowledge of scientific methods, experimental approaches, and data analysis techniques is no less important than mastering formulas, and enables practical application of physics.
The high school course includes mechanics, molecular physics and thermodynamics, electrodynamics, oscillations and waves, optics, quantum and atomic physics. The curriculum is structured hierarchically: from simple mechanical phenomena to complex quantum processes. There are standard and advanced levels with varying degrees of material detail.
First, formulate your objective and hypothesis, prepare equipment, and study the measurement methodology. Conduct measurements accounting for uncertainties, and record data in a table. Process the results, construct graphs, calculate uncertainties, and draw conclusions about confirming or refuting the hypothesis.
Molecular physics studies the properties of matter based on its molecular structure and kinetic theory. It explains the behavior of gases, liquids, and solids through the motion and interaction of molecules. This section is closely connected to thermodynamics and explains thermal phenomena at the microscopic level.
Historical context shows the development of scientific ideas and methods for understanding nature. Studying the contributions of outstanding physicists and engineers helps understand the logic of scientific discoveries and forms a scientific worldview. History demonstrates that science is a living process, not a set of ready-made truths.
Advanced level involves greater theoretical detail, complex problems, and expanded experimental work. Standard level provides a general understanding of physical phenomena and basic laws. Advanced study prepares students for admission to technical universities and scientific careers.
No, classical mechanics is a necessary foundation for studying quantum physics. Quantum phenomena are often explained through contrast with classical concepts. Without understanding Newton's laws, energy, and momentum, it's impossible to grasp quantum concepts and their revolutionary nature.
Physical principles underlie all technologies—from electronics to spacecraft. Understanding electromagnetism is essential for developing communications, quantum physics for creating semiconductors and lasers. Thermodynamics applies to energy systems, while mechanics drives mechanical engineering and transportation.
No, that's a myth. Modern physics is actively developing: researchers are investigating dark matter, quantum computers, fusion energy, and nanomaterials. Many fundamental questions remain open, such as unifying quantum mechanics and gravity. Physics continues to discover new phenomena and create breakthrough technologies.
The scientific method is a systematic approach to knowledge: observation, hypothesis formulation, experimentation, data analysis, and conclusions. In physics, it includes mathematical modeling and testing theoretical predictions through experiment. This method ensures objectivity and reproducibility of scientific results.
Organize measurements in tables, calculate mean values and uncertainties. Plot graphs of relationships, determine the nature of connections between quantities. Compare results with theoretical predictions, assess reliability, and formulate conclusions considering the limits of applicability.
There are national education standards that define the required minimum content. However, schools can choose textbooks and programs that differ in topic sequence and depth of study. Importantly, all programs include theory, experimentation, and methodological preparation at either foundational or advanced levels.
