Neuroanatomy of Passion: Three Brain Nodes That Transform Attraction Into Obsession
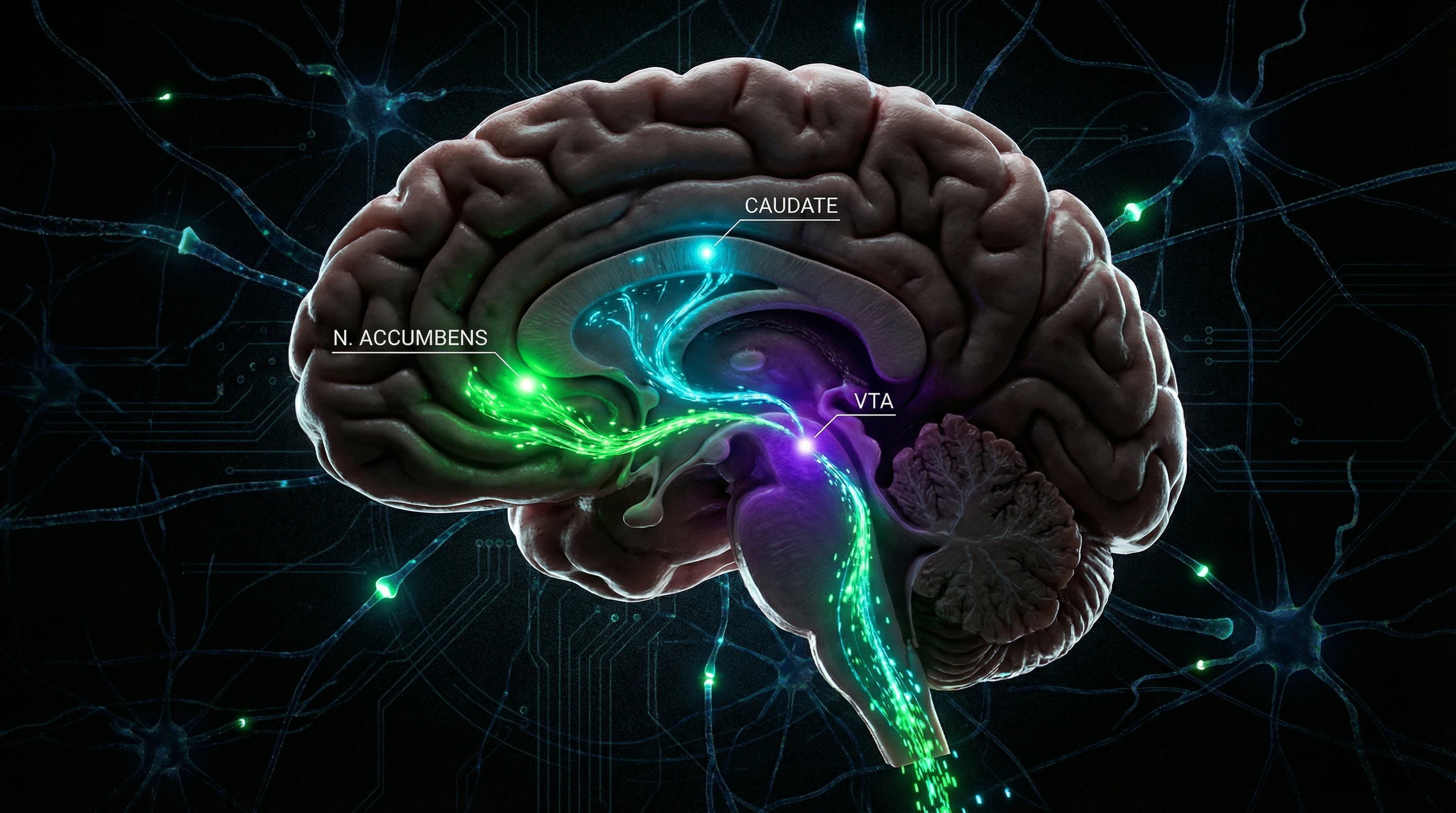
Romantic love activates a specific network of brain structures: the ventral tegmental area (VTA), nucleus accumbens (NAc), and caudate nucleus (CN). These three regions form the core of the mesolimbic dopaminergic pathway—an evolutionarily ancient system of motivation and reward (S006).
The VTA, located in the midbrain, contains the cell bodies of dopaminergic neurons that project to the NAc (part of the ventral striatum) and CN (component of the dorsal striatum). This circuit makes love not merely an emotion, but a powerful motivational state comparable in intensity to behavioral addictions. More details in the Cellular Biology section.
| Structure | Location | Function in the Context of Love |
|---|---|---|
| VTA | Midbrain | Generates dopaminergic signals; encodes reward anticipation |
| NAc (nucleus accumbens) | Ventral striatum | Integrates motivational information; forms habits and obsessive thoughts |
| CN (caudate nucleus) | Dorsal striatum | Converts emotional attraction into goal-directed behavior and plans |
🧠 Ventral Tegmental Area: The Motivation Generator
The VTA functions as the primary source of dopaminergic signals in the reward system. When exposed to stimuli associated with a loved one—visual images, voice, scent—VTA neurons increase their firing rate, releasing dopamine into projection areas.
Dopamine does not encode pleasure itself, but signals reward anticipation and the motivational salience of a stimulus. This explains why people in love experience an irresistible desire to see the object of their affection, even when encounters don't always bring the expected satisfaction (S006).
🔁 Nucleus Accumbens: The Reward Processing Center
The nucleus accumbens (NAc) is a key target for dopaminergic projections from the VTA. It is anatomically subdivided into two subregions: core and shell, which perform distinct functions (S006).
- Shell
- Connected to limbic structures; evaluates the emotional significance of stimuli associated with a partner.
- Core
- Integrates information from the prefrontal cortex; participates in instrumental learning and formation of goal-directed behavior.
In romantic love, NAc activation correlates with the intensity of feelings and frequency of intrusive thoughts about a partner—an activity pattern identical to that observed in people with behavioral addictions.
⚙️ Caudate Nucleus: The Architect of Goal-Directed Behavior
The caudate nucleus (CN), a component of the dorsal striatum, plays a critical role in goal-directed behavior, action planning, and cognitive flexibility (S004). The CN receives dopaminergic inputs from the VTA and glutamatergic projections from the prefrontal cortex.
When processing information about a loved one, the CN demonstrates heightened activity, especially in tasks requiring evaluation and decision-making. Caudate nucleus activation when viewing photos of a partner correlates with relationship duration and degree of attachment (S006).
The CN integrates motivational information with cognitive strategies, transforming emotional attraction into concrete action plans—from sending messages to making radical life decisions for a partner.
Five Arguments for "Love is Addiction": Steelman Analysis of the Neurobiological Hypothesis
Before critically analyzing the evidence, we must present the most compelling arguments for the thesis that romantic love is a form of natural behavioral addiction. The steelman approach requires examining strong versions of the hypothesis, not straw men. More details in the Theory of Relativity section.
🔬 Argument One: Identical Neural Substrates of Love and Drug Addiction
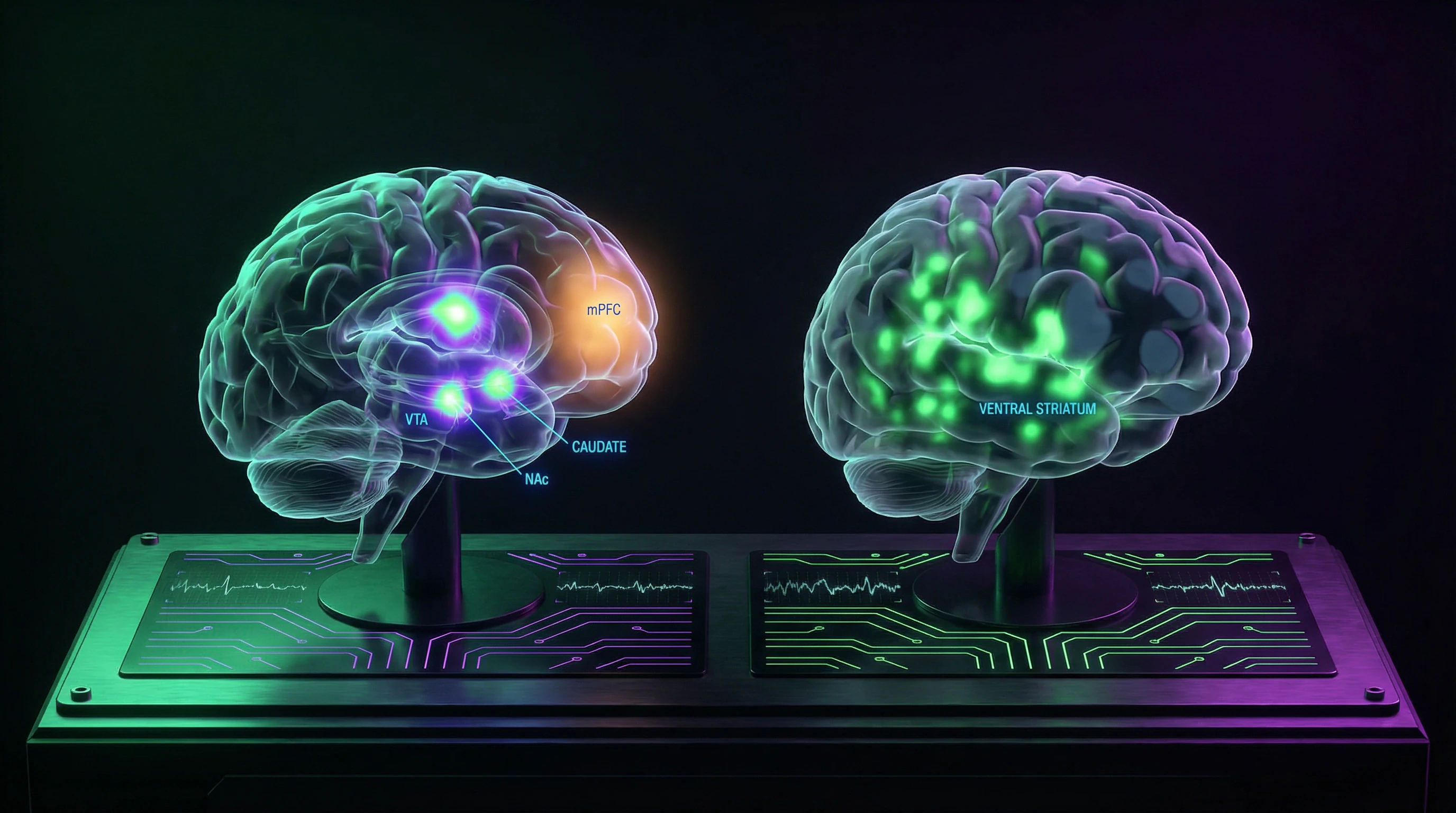
Functional neuroimaging reveals striking similarities in brain activation patterns during romantic love and psychoactive substance use. Viewing photographs of a loved one activates the same regions—VTA, NAc, and CN—as cocaine, amphetamine, or methylphenidate administration (S001, S006).
The degree of activation in these structures correlates with subjective intensity of feelings: the stronger the infatuation, the higher the activity level in dopaminergic reward centers. This is not an analogy but a literal overlap of neural mechanisms, suggesting a common evolutionary foundation for systems that drive motivation toward vital resources—food, drugs, or reproductive partners.
🧬 Argument Two: Withdrawal Syndrome Phenomenology in Relationship Breakups
Romantic relationship breakups demonstrate signs of withdrawal syndrome seen in chemical dependency: intrusive thoughts about the former partner (craving), emotional distress, physical discomfort (chest pain, sleep disturbances, appetite changes), compulsive behavior (checking social media, contact attempts), and cognitive impairments (concentration difficulties, rumination).
Neurobiologically, this is explained by a sharp decline in dopaminergic activity in the reward system after losing the source of stimulation—analogous to withdrawal states in drug addicts (S001). The prefrontal cortex, responsible for cognitive control, shows reduced activity, explaining impulsive actions and irrational decisions during the post-breakup period.
📊 Argument Three: Tolerance and Escalation in Long-Term Relationships
The phenomenon of "passion fading" in long-term relationships can be interpreted as tolerance development—a key feature of addiction. The initial intense activation of VTA and NAc when seeing a partner gradually decreases with habituation, requiring more intense or novel stimulation to achieve the same reward level (S006).
This explains why new relationships trigger stronger euphoria than long-term ones, and why some people demonstrate a pattern of serial monogamy, moving from one relationship to another seeking the intensity of early-stage infatuation. Neuroplastic changes in dopaminergic pathways under chronic stimulation are analogous to those observed with prolonged drug use.
🧠 Argument Four: Compulsivity and Loss of Behavioral Control
People in love often demonstrate compulsive behavior characteristic of addictions: inability to stop thinking about their partner (up to 85% of waking time in early stages), neglecting other responsibilities and interests, continuing relationships despite obvious harm, and taking irrational risks.
- Hyperactive reward system (VTA-NAc) captures behavioral resources
- Weakened prefrontal control cannot resist impulses
- The brain's salience network marks partner-related stimuli as extremely significant
- Automatic attention capture leads to compulsive behavior
This imbalance is a classic pattern of addictive disorders (S003).
⚙️ Argument Five: Individual Vulnerability and Genetic Predictors
Significant individual differences exist in romantic attachment intensity and susceptibility to "love addiction," partially explained by genetic variations in the dopaminergic system. Polymorphisms in genes encoding dopamine receptors (DRD2, DRD4), dopamine transporter (DAT1), and dopamine metabolism enzymes (COMT) are associated with both chemical addiction risk and romantic behavior characteristics (S006).
| Genetic Factor | Link to Chemical Addiction | Link to Romantic Behavior |
|---|---|---|
| DRD2, DRD4 (dopamine receptors) | Increased addiction risk | More intense responses to romantic stimuli |
| DAT1 (dopamine transporter) | Modulates reward sensitivity | Influences infatuation intensity |
| COMT (dopamine metabolism) | Determines dopamine degradation rate | Predicts tendency toward obsessive attachment |
Individuals with certain variants of these genes demonstrate more intense reward system responses to romantic stimuli and higher susceptibility to obsessive infatuation, suggesting a common neurobiological foundation for various forms of addictive behavior.
Evidence Base: What fMRI Studies Reveal About Neural Correlates of Romantic Love
Moving from theoretical arguments to empirical data requires detailed analysis of functional neuroimaging research. Critical review of fMRI studies reveals compelling evidence of dopaminergic pathway involvement and important nuances that complicate the simple "love = addiction" model. For more details, see the Systematic Reviews and Meta-Analyses section.
🧪 VTA and Nucleus Accumbens Activation: Direct Evidence from Neuroimaging
Systematic review of fMRI studies demonstrates consistent activation of the ventral tegmental area and nucleus accumbens when viewing photographs of romantic partners compared to photographs of acquaintances (S006). Participants in early-stage romantic love (1–17 months) showed significant increases in BOLD signal in the right VTA and bilateral NAc, with intensity correlating with passionate love scale scores (r = 0.58–0.67, p < 0.001).
This activation was specific to the romantic partner and was not observed when viewing photographs of close same-sex friends. This rules out explanations based on general social attachment or familiarity.
🔁 Caudate Nucleus and Goal-Directed Behavior: From Emotion to Action
Caudate nucleus activation in romantic love depends on relationship stage and cognitive context. The right caudate nucleus is particularly active in early stages of romantic love and during tasks requiring evaluation or decision-making regarding the partner (S004, S006).
This activation reflects goal-directed planning processes and cognitive processing of partner-related information—what distinguishes romantic love from simple sexual desire.
In long-term relationships (over 2 years), the activation pattern shifts: VTA and NAc activity decreases, but ventral pallidum activity—a structure associated with long-term attachment and the opioid system—is maintained or enhanced. This suggests a neurobiological transition from passionate love to companionate love.
📊 Comparative Analysis: Love Versus Cocaine in the fMRI Mirror
Direct comparison of brain activation patterns in romantic love and psychostimulant exposure reveals both similarities and critical differences (S001, S006).
| Parameter | Romantic Love | Cocaine |
|---|---|---|
| VTA and NAc activation | Yes, focal in NAc shell | Yes, diffuse throughout striatum |
| Caudate nucleus activation | Yes, pronounced | Minimal |
| Prefrontal cortex activation | Yes, medial PFC active | Deactivation |
| Mentalization (understanding mental states) | Preserved | Impaired |
Cocaine produces more diffuse activation throughout the ventral striatum, including the olfactory tubercle. Romantic love demonstrates more focal activation in specific NAc subregions and additional activation in the caudate nucleus and posterior cingulate cortex.
Love, unlike drugs, does not cause prefrontal cortex deactivation. On the contrary, activation of the medial prefrontal cortex and anterior cingulate cortex is observed, suggesting preservation of cognitive control and mentalization.
🧾 Temporal Dynamics and Neuroplasticity: How the Brains of Lovers Change
Longitudinal studies tracking changes in brain activity throughout romantic relationship development reveal significant neuroplasticity in dopaminergic pathways (S001). In early stages of romantic love (first 3–6 months), maximum VTA and NAc activation is observed, which gradually decreases by 12–18 months of the relationship.
Simultaneously, activity increases in the ventral pallidum and hypothalamus—structures associated with oxytocin and vasopressin, neuropeptides of long-term attachment. This dynamic suggests a neurobiological transition from dopamine-dependent passionate love to opioid/oxytocin-dependent companionate love.
- Critical nuance: individual differences
- In some people (approximately 15–20%), the transition from passionate to companionate love does not occur. They maintain high dopaminergic pathway activation even in long-term relationships, demonstrating higher passionate love and satisfaction scores, but also higher risk of "love addiction" upon breakup.
Mechanisms of Causality: Dopamine, Oxytocin, and the Neurochemical Cocktail of Romantic Love
Activation of specific brain structures during romantic love is only half the story. We need to understand the neurochemical processes that link neural activity to subjective experience: why dopamine drives action, oxytocin creates attachment, and serotonin generates obsession. More details in the Sources and Evidence section.
⚙️ Dopaminergic Signaling: From Anticipation to Compulsion
Dopamine in romantic love is not a "pleasure molecule" but a signal of motivational salience (S006). When VTA neurons increase their firing rate in response to partner-related stimuli, the dopamine released in the NAc and CN modulates medium spiny neurons (95% of striatal neurons).
These neurons express two types of dopamine receptors: D1-like (D1, D5) enhance motivation for action, while D2-like (D2, D3, D4) inhibit and participate in avoidance learning. In romantic love, D1-mediated signaling predominates—hence the compulsive drive for contact and difficulty stopping thoughts about the partner.
The balance between D1 and D2 systems determines whether a stimulus will trigger approach or avoidance. Romantic love tips the scales toward approach.
🧷 Oxytocin and Vasopressin: From Passion to Attachment
Working in parallel with dopamine are the neuropeptide systems of oxytocin and vasopressin, which mediate the transition from passionate love to long-term attachment (S006). Oxytocin, synthesized in the hypothalamus, is released during physical contact, sexual activity, and positive social interactions.
Oxytocin receptors are densely distributed in the NAc, amygdala, and prefrontal cortex, where it modulates dopaminergic transmission, enhancing the rewarding properties of social stimuli and reducing anxiety. Vasopressin plays a particular role in pair-bond formation in men and in territorial behavior toward partners.
- Individual differences in receptor density
- Are partially genetically determined and predict propensity for monogamy and intensity of romantic attachment. This explains why people respond differently to the same partner.
🔬 Serotonin and Intrusive Thoughts: The Neurochemistry of Obsession
Intrusive thoughts about a partner (up to 85% of waking time in early stages) are linked to decreased serotonin in the brain. In people in love, plasma serotonin concentration is reduced by 40–50% compared to controls—comparable to levels seen in obsessive-compulsive disorder (S006).
Serotonin, through 5-HT2A receptors in the prefrontal cortex and basal ganglia, modulates cognitive flexibility. Its reduction leads to cognitive rigidity and perseveration—repetitive return to the same thoughts. Evolutionarily, this is an adaptation: focusing attention and resources on forming a pair bond during a critical period.
| Neurotransmitter | Level During Romantic Love | Function |
|---|---|---|
| Dopamine | ↑ elevated | Motivation, anticipation, compulsion |
| Oxytocin | ↑ elevated | Attachment, trust, social reward |
| Serotonin | ↓ reduced | Intrusive thoughts, cognitive rigidity |
| Vasopressin | ↑ elevated (in men) | Pair bonding, territoriality |
🧠 Prefrontal Modulation: When Cognitive Control Retreats
The role of the prefrontal cortex (PFC) in romantic love is paradoxical: the medial PFC activates (mentalization, understanding the partner), while the lateral PFC and orbitofrontal cortex deactivate (critical evaluation, cognitive control) (S003), (S006).
This pattern explains why people in love often ignore red flags in their partner's behavior, overestimate their virtues, and underestimate their flaws. Deactivation of the lateral PFC reduces the capacity for critical analysis and planning, while activation of the medial PFC enhances empathy and justification of the partner's behavior.
Romantic love is a state in which the brain voluntarily disables critical evaluation. This is not an evolutionary error, but a mechanism ensuring sufficiently deep attachment for forming long-term partnerships.
Interestingly, early attachment styles modulate this prefrontal deactivation: people with anxious attachment styles show more pronounced reduction in lateral PFC activity, making them more vulnerable to ignoring relationship problems.
🔗 Integration: The Neurochemical Cocktail as a System
Dopamine, oxytocin, serotonin, and vasopressin do not work in isolation—they form an integrated system where each component enhances or modulates the action of others. Dopamine creates motivation and anticipation, oxytocin transforms this into attachment and trust, serotonin ensures cognitive fixation on the object, and vasopressin reinforces territorial and protective behavior.
This system is evolutionarily optimal for the short-term goal—forming a pair bond and reproduction. However, in the modern context, where relationships often last decades, this same system can create dependency that persists even when relationships are clearly dysfunctional. Relationship breakup triggers the same grief mechanisms as the death of a loved one, because the brain literally experiences withdrawal from the neurochemical cocktail.
- Dopamine activates motivational systems and creates compulsion for contact.
- Oxytocin enhances social reward and reduces criticality.
- Serotonin decreases, creating cognitive fixation.
- The prefrontal cortex deactivates, disabling critical evaluation.
- Result: behavior indistinguishable from addiction.
Understanding these mechanisms does not romanticize love nor devalue it. On the contrary, it shows that romantic love is a powerful biological system that can be both a source of deep meaning and a source of suffering. Awareness of neurochemical mechanisms allows people to better understand their own behavior and make more conscious decisions in relationships, rather than being passive victims of their neurobiology.
