What is neuropeptide regulation of attachment — and why it's not just "chemistry of love"
Attachment in the neurobiological sense is a stable emotional bond between individuals, mediated by specific brain systems and neurochemical pathways. Formation and maintenance of attachment requires coordinated activity of multiple neuropeptide and neurotransmitter systems (S004, S006), not simply "love hormones."
Oxytocin and vasopressin play central roles, but do not function in isolation — they operate in tight integration with opioid, noradrenergic, dopaminergic, and serotonergic pathways. More details in the Quantum Mechanics section.
🧬 Molecular architecture: two neuropeptides, nine amino acids
Oxytocin and vasopressin are evolutionarily ancient neuropeptides, differing by only two amino acids out of nine. This minimal structural difference leads to substantial functional differences.
| Neuropeptide | Primary function | Activation context |
|---|---|---|
| Oxytocin | Affiliative behavior, maternal care, social trust | Proximity, social contact |
| Vasopressin | Social recognition, territorial behavior, pair bonding (males) | Social competition, partner defense |
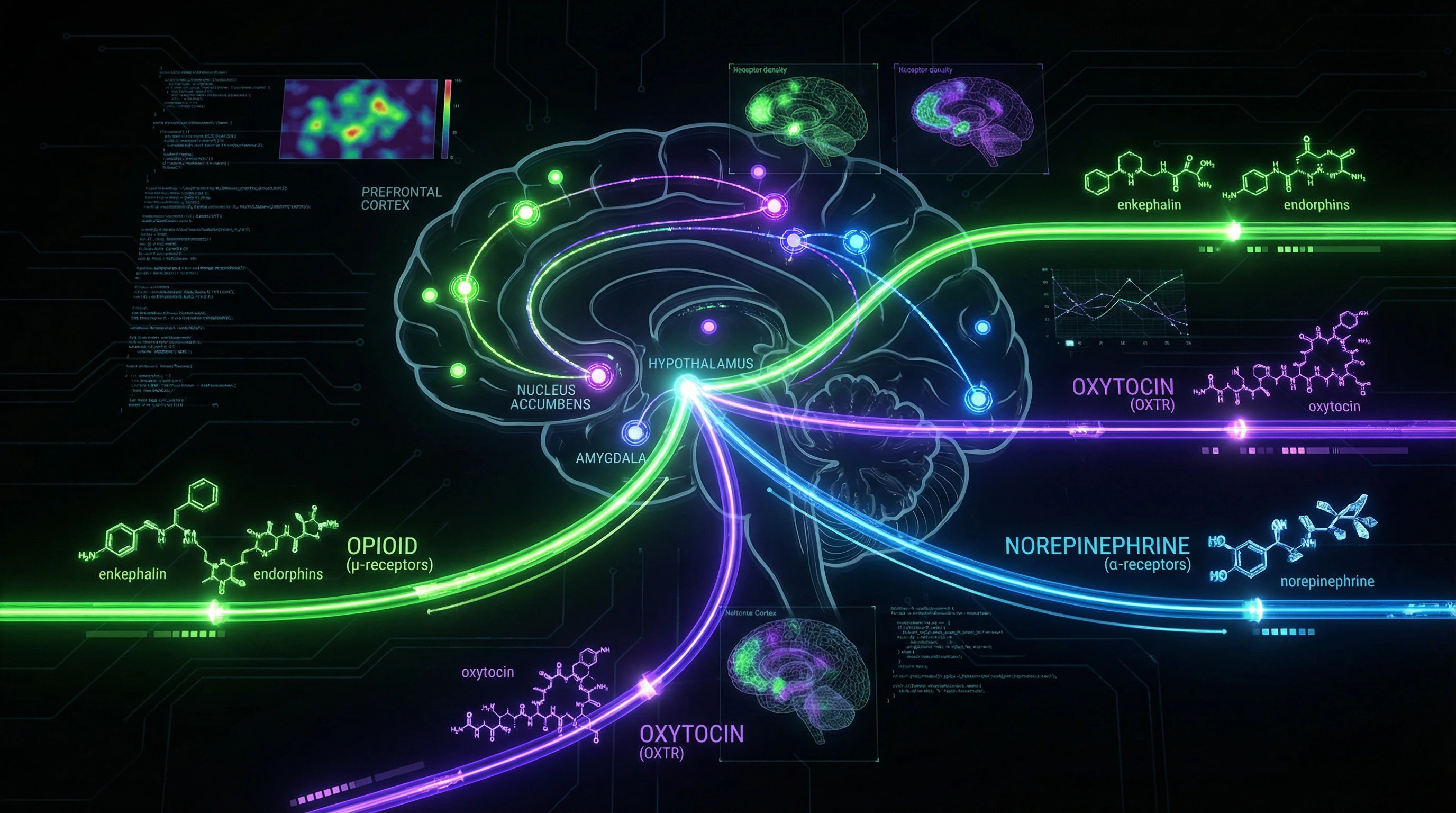
Both are synthesized in the paraventricular and supraoptic nuclei of the hypothalamus, from where they are released both into the bloodstream (acting as hormones) and into various brain regions (acting as neuromodulators) (S001, S002).
🔁 Receptor specificity: why one neuropeptide — different effects
The effects of oxytocin and vasopressin are determined not only by their concentration, but also by the distribution of receptors in the brain, which varies between species, sexes, and individuals (S006, S008).
- Oxytocin receptors (OXTR)
- High density in nucleus accumbens, amygdala, prefrontal cortex. Activation → social trust, reduced anxiety.
- Vasopressin receptors (V1aR, V1bR)
- Distributed in hippocampus, septum, amygdala. Activation → social recognition, aggression in territorial defense.
The same neuropeptide can produce opposite behavioral effects depending on which neuronal populations are activated — a phenomenon that completely contradicts the simplified notion of "oxytocin as the love hormone" (S008).
🧱 Three neurochemical systems: opioids, oxytocin, noradrenaline
Formation of stable emotional bonds requires sequential and parallel activation of three primary neurochemical systems (S002).
- Opioid system: μ-opioid receptors create sensations of pleasure and comfort in proximity to the attachment figure.
- Oxytocin system: modulates social recognition and reduces anxiety in the presence of a familiar individual.
- Noradrenergic system: activated during separation, creating an aversive state that motivates restoration of contact.
This three-component architecture explains why attachment involves not only positive reinforcement of proximity, but also negative reinforcement of separation. Breaking the bond activates the same stress systems as physical pain — this is not a metaphor, but a neurobiological fact.
Iron Man: Seven Arguments for the Central Role of Oxytocin and Vasopressin
Before analyzing the limitations, it is necessary to present the most compelling evidence for the fundamental role of oxytocin and vasopressin in regulating attachment and social behavior. These arguments are based on convergent data from comparative neurobiology, pharmacological manipulations, genetic studies, and neuroimaging. For more details, see the section Systematic Reviews and Meta-Analyses.
🔬 Argument One: Species-Specific Variations in Social Organization Correlate with Receptor Patterns
Classic studies on voles (Microtus) have demonstrated a striking connection between a species' social organization and the distribution of vasopressin and oxytocin receptors in the brain (S002, S006). Monogamous prairie voles (M. ochrogaster) exhibit high density of V1aR in the ventral pallidum and OXTR in the nucleus accumbens, whereas promiscuous montane voles (M. montanus) have substantially lower density of these receptors in the same regions.
Experimental increase in V1aR expression in the ventral pallidum of promiscuous voles induces pair-bonding behavior characteristic of monogamous species (S006). This demonstrates not merely correlation, but a causal relationship between neuropeptide systems and the formation of stable social bonds.
The molecular architecture of receptors determines the social architecture of a species — and this can be experimentally rewritten.
🧪 Argument Two: Pharmacological Blockade Disrupts Attachment Formation
Administration of oxytocin or vasopressin receptor antagonists during critical periods prevents the formation of normal attachment (S004, S006). In rat pups, systemic administration of an oxytocin receptor antagonist disrupts the formation of maternal odor preference, even when all other aspects of maternal care are preserved.
In adult prairie voles, V1aR blockade prevents pair-bond formation after mating, despite normal sexual behavior. Oxytocin and vasopressin do not merely accompany attachment — they are necessary for its formation.
📊 Argument Three: Exogenous Administration Accelerates Social Bond Formation
Administration of oxytocin or vasopressin can accelerate or enhance attachment formation (S001, S002). In prairie voles, central administration of oxytocin induces pair-bond formation even without mating, if the female is simply in the presence of a male.
In humans, intranasal oxytocin administration enhances trust in economic games, improves emotion recognition from facial expressions, and increases attention to social stimuli (S008). While these effects are context-dependent and not universal, they confirm the modulatory role of oxytocin in social behavior.
🧬 Argument Four: Genetic Variations in Receptor Genes Are Linked to Individual Differences
Polymorphisms in the OXTR and AVPR1A genes are associated with individual differences in social behavior and attachment in humans (S008). Certain OXTR variants are linked to differences in maternal sensitivity, empathy, social anxiety, and even autism risk.
Variations in the AVPR1A promoter region correlate with the quality of pair relationships in men and the likelihood of divorce. Although the effect sizes of these genetic variations are relatively small, their consistency across multiple studies confirms the role of these neuropeptide systems in shaping individual attachment trajectories. For more on genetic mechanisms, see the AVPR1A gene analysis.
🧠 Argument Five: Neuroimaging Shows Activation of Specific Brain Networks
fMRI studies demonstrate that oxytocin modulates activity in key social brain regions: the amygdala, anterior cingulate cortex, insula, and medial prefrontal cortex (S008). When viewing faces of loved ones or infants, activation of oxytocin-sensitive regions is observed, with the activation pattern correlating with subjective ratings of attachment.
Administration of exogenous oxytocin alters functional connectivity between these regions, strengthening coordination between reward and emotional regulation systems (S005).
🔁 Argument Six: Early Experience Modulates System Sensitivity Through Epigenetic Mechanisms
The quality of early maternal care influences oxytocin receptor expression through epigenetic mechanisms, such as DNA methylation in the OXTR gene promoter region. Rat pups receiving high levels of maternal licking and grooming demonstrate increased OXTR expression in the amygdala and lower anxiety in adulthood.
In humans, childhood experiences of abuse or neglect are associated with altered OXTR methylation and impairments in social functioning. The oxytocin system is not simply innate — it is plastic and shaped by early social experience. The mechanisms of this plasticity are examined in the study on the neurobiology of attachment styles.
⚙️ Argument Seven: Evolutionary Conservation of Systems Across Mammalian Taxa
Oxytocin and vasopressin (or their homologs) are found in all mammals and even in more ancient vertebrates, where they regulate social and reproductive behavior (S001). This evolutionary conservation points to the fundamental role of these neuropeptides in social organization.
Specific functions and receptor expression patterns vary depending on a species' social structure, demonstrating how evolution uses conserved molecular tools to create diverse social systems (S006).
- Species-specific receptor variations correlate with social organization and can be experimentally rewritten.
- Pharmacological blockade disrupts attachment; exogenous administration accelerates it.
- Genetic polymorphisms in OXTR and AVPR1A are linked to individual differences in social behavior.
- Neuroimaging shows activation of specific brain networks in response to social stimuli.
- Early experience modulates system sensitivity through epigenetic mechanisms.
- Evolutionary conservation points to the fundamental role of these neuropeptides.
Evidence Base: What the Data Shows About Mechanisms of Action and Limitations
Moving from general arguments to specifics, we need to examine the concrete mechanisms through which oxytocin and vasopressin influence attachment, as well as critical limitations in existing data. The scientific literature demonstrates a significantly more complex picture than popular simplifications. For more details, see the Scientific Databases section.
🧪 Multiple Neurotransmitter Systems Work in Coordination, Not Isolation
A key misconception is the notion that oxytocin and vasopressin act as the sole or even primary regulators of attachment. Attachment formation requires coordinated activity of at least three major neurochemical systems: opioid, oxytocinergic, and noradrenergic (S004).
The opioid system, acting through μ-opioid receptors in the nucleus accumbens and ventral tegmental area, provides immediate positive reinforcement of social contact. Blockade of opioid receptors with naloxone disrupts maternal attachment formation in rat pups as effectively as blockade of oxytocin receptors.
| System | Mechanism | Function in Attachment |
|---|---|---|
| Opioid | μ-receptors in nucleus accumbens | Positive reinforcement of contact |
| Noradrenergic | α2-adrenoreceptors in locus coeruleus | Separation distress, motivation to restore contact |
| Dopaminergic | Ventral striatum | Signal integration, predictive value of social stimuli |
The noradrenergic system plays a critical role in creating an aversive state during separation from the attachment figure. Activation of α2-adrenoreceptors during separation creates distress that motivates contact restoration. This explains why attachment involves not only seeking proximity but also avoiding separation.
📊 Context-Dependent Effects of Oxytocin: Not a Universal Prosocial Agent
Critical analysis of the human oxytocin literature reveals substantial context-dependence of its effects, contradicting the simple "love hormone" narrative (S008). Oxytocin enhances trust and cooperation toward in-group members but can simultaneously increase distrust and defensive aggression toward outsiders.
Oxytocin modulates social salience and attention to social cues rather than simply inducing prosociality. Its effects depend on individual differences in baseline anxiety, attachment style, and social experience.
In economic games, oxytocin increases generosity toward partners perceived as in-group members but doesn't affect or even reduces cooperation with out-group members. In individuals with avoidant attachment styles, oxytocin can increase rather than decrease social anxiety. In people with histories of social trauma, oxytocin can activate negative social memories.
These data demonstrate that oxytocin doesn't have a fixed behavioral effect but modulates social information processing in ways dependent on context and individual history. For more on how early experience reprograms these systems, see the analysis of the neurobiology of attachment styles.
🧬 Species-Specific Variations Limit Direct Translation from Animal Models to Humans
While the basic neurochemistry of oxytocin and vasopressin is conserved across species, specific patterns of receptor expression and behavioral effects vary substantially (S006). Classic vole studies demonstrating the role of V1aR in pair bond formation cannot be directly extrapolated to primates, which have different vasopressin receptor distributions.
- Voles
- Vasopressin is critical for pair bonding; pronounced sex differences in neuropeptide functions.
- Primates (including humans)
- Role of vasopressin in pair relationships is less clear; sex differences are less pronounced; developed prefrontal cortex provides cognitive regulation partially independent of basic neuropeptide systems.
In humans, attachment involves additional levels of cognitive processing—mentalization, narrative construction of relationships, conscious emotion regulation—that aren't fully captured by rodent-based models. This is particularly important when analyzing why ideas about a "genetic fidelity switch" remain a scientific illusion: see the critique of the AVPR1a gene.
🔁 Interaction with the HPA Axis: Stress Modulates Neuropeptide Systems
A critically important aspect often overlooked in popular accounts is the interaction of the oxytocin system with the hypothalamic-pituitary-adrenal (HPA) axis, which regulates stress responses (S004). Oxytocin exerts anxiolytic effects partly through suppression of HPA axis activity, reducing release of corticotropin-releasing hormone (CRH) and subsequent cortisol secretion.
However, this connection is bidirectional: chronic stress and elevated cortisol can disrupt oxytocin system functioning. Early life stress, such as maternal deprivation or unpredictable maternal care, leads to long-term changes in HPA axis sensitivity and the oxytocin system.
- Animals experiencing early stress show reduced oxytocin receptor expression in key brain regions.
- Elevated baseline HPA axis activity and disrupted attachment patterns in adulthood.
- In humans, childhood abuse is associated with altered oxytocin system reactivity and increased risk of disorganized attachment.
The oxytocin system doesn't function in isolation but is deeply integrated with stress regulation systems. This explains why relationship breakups trigger the same grief mechanisms as the death of a loved one.
🧠 Brain Network Dynamics: Oxytocin Modulates Functional Connectivity
Contemporary approaches are shifting from focusing on individual brain regions to analyzing how oxytocin modulates functional connectivity between regions (S005). Research shows that oxytocin enhances coordination between social brain areas, increasing synchrony of their activity.
Oxytocin strengthens connectivity between the amygdala and prefrontal cortex, explaining its anxiolytic effects through enhanced top-down regulation of emotional responses.
Oxytocin also modulates the balance between the default mode network and networks oriented toward external stimuli. During social interaction, oxytocin can enhance attentional switching to social stimuli through modulation of the salience network.
These effects at the level of large-scale brain networks explain why oxytocin influences not specific behavioral acts but the general orientation of attention and information processing in social contexts.

Mechanisms of Causality: How to Distinguish Correlation from Causation in Neuropeptide Research
One of the central questions in interpreting data about oxytocin and vasopressin is distinguishing between correlation and causation. The fact that oxytocin levels rise during social interaction doesn't necessarily mean that oxytocin causes attachment—it may be a consequence rather than a cause of social behavior. For more details, see the Sources and Evidence section.
🔬 Experimental Manipulations: The Gold Standard for Establishing Causality
The most convincing evidence for the causal role of oxytocin and vasopressin comes from experiments with pharmacological manipulations (S001, S004, S006). Administration of receptor antagonists (blocking neuropeptide action) or agonists (mimicking their action) allows us to establish whether these systems are necessary and sufficient for attachment formation.
Blockade of oxytocin receptors disrupts maternal attachment formation in rat pups, while oxytocin administration accelerates pair bond formation in voles even without mating. But the effects are nonlinear: low doses often produce opposite effects to high doses.
Peripheral administration (intranasal in humans) has different effects than central administration directly into the brain. The timing of administration relative to social interaction is critical: oxytocin before interaction enhances attention to social stimuli, after—affects consolidation of memory about the partner.
This means that the same neuropeptide can act at different stages of the social process through different mechanisms. Research on animal models (especially voles and primates) provides more direct evidence of causality than correlational studies in humans.
⚠️ Interpretation Pitfalls: Why "Brain Correlation" Doesn't Equal "Behavioral Cause"
Even when we see activation of specific neural circuits during social behavior, this doesn't prove that this activation causes the behavior. Neural activity may be an epiphenomenon—a byproduct rather than a driving force.
| Type of Evidence | Strength of Causal Inference | Main Pitfall |
|---|---|---|
| Correlation of neuropeptide levels with behavior | Weak | Reverse causality: behavior triggers release, not vice versa |
| Neuroimaging of activation during social interaction | Weak–moderate | Activation may be consequence, not cause; multiple systems active simultaneously |
| Pharmacological manipulation (administration/blockade) in animals | Strong | Doses and routes of administration may not match physiological conditions |
| Genetic manipulation (knockouts, knockdowns) | Strong | Compensatory mechanisms; developmental effects may mask acute effects |
| Intranasal oxytocin administration in humans | Moderate | Unclear whether drug reaches brain; placebo effect; inter-individual variability |
Intranasal oxytocin administration in humans is a popular method, but its interpretation requires caution. It's unclear to what extent the drug reaches the central nervous system, and effects are often modest and inconsistent (S007, S008).
Additionally, the neurobiology of attachment styles shows that the same neuropeptide can have opposite effects depending on developmental history and context. Oxytocin can enhance trust toward in-group members while simultaneously reducing trust toward outsiders—this isn't a contradiction, but evidence that the system operates within the context of social categorization.
🔄 Multiplicity of Mechanisms: Why One Neuropeptide Doesn't Equal One Function
Oxytocin and vasopressin act through multiple receptors in different brain structures. The same neuropeptide can simultaneously enhance partner attachment, reduce anxiety, modulate pain sensitivity, and influence feeding behavior.
- Oxytocin in the amygdala reduces fear of social stimuli
- Oxytocin in the hypothalamus modulates sexual and maternal behavior
- Oxytocin in the striatum is linked to reward from social contact
- Vasopressin in the septum and amygdala regulates aggression and territorial behavior
- Vasopressin in the hypothalamus affects water-electrolyte balance and osmotic regulation
This means that blocking one receptor can have multiple side effects, and interpreting results requires understanding the entire system, not just an isolated component.
Causality in neurobiology is not a binary property ("present" or "absent"), but a spectrum of probabilities depending on context, dose, timing, and individual differences. Even the most convincing experimental manipulations don't guarantee that the mechanism works the same way in humans under natural conditions.
Research on the AVPR1a gene and monogamy demonstrates this problem: gene polymorphism is associated with variability in social behavior, but the effect size is small, and the mechanism remains unclear. Genetic association is not the same as mechanistic explanation.
📊 Criteria for Evaluating Causality in Neuropeptide Research
When evaluating a study, ask yourself: was there control for confounders (variables that could explain the result)? Were there blind procedures (researcher didn't know who received the drug)? Are the results reproducible across different laboratories and species?
Human studies often rely on self-reports of feelings and behavior, which are subject to social desirability and expectations. Even when objective measures are used (e.g., choices in economic games), interpretation requires caution: choices may result from multiple factors, not just oxytocin.
The most reliable conclusions about causality come from combining methods: pharmacological manipulations in animal models, genetic studies in humans, neuroimaging, and behavioral tests. No single method is sufficient (S001, S002).
