What a neurointerface actually is: from electrical brain patterns to robot commands
A brain-computer interface is a direct communication channel between the electrical activity of the brain and an external device: a computer, prosthetic, exoskeleton, or communication system (S001). The key word is "direct": the technology bypasses traditional command pathways through the peripheral nervous system and muscles.
Instead of pressing a button with your hand, the user generates a specific pattern of brain activity that the system recognizes and converts into a control signal. This isn't telepathy or mind reading — it's decoding the electrical codes the brain already uses to control the body. More details in the Abiogenesis section.
Three levels of definition
- Technical level
- Registration of bioelectrical brain signals (most commonly through electroencephalography), their digital processing using machine learning algorithms, and conversion into commands for executive devices.
- Neurophysiological level
- Use of specific components of brain electrical activity: the P300 wave (occurring 300 milliseconds after a significant stimulus), visual evoked potentials (VEP) in response to visual stimulation, or sensorimotor rhythm patterns associated with imagined movement (S003).
- Functional level
- A tool for restoring lost communicative or motor functions, expanding neurorehabilitation capabilities, and creating new forms of human interaction with technological systems (S004).
Boundaries of applicability
BCI does not include systems using electromyography (recording muscle activity) — that's no longer a direct channel from the brain. Eye-tracking systems also don't qualify as BCI, though they're often used in conjunction.
Neurointerfaces differ from deep brain stimulation (DBS) systems, which transmit signals in the opposite direction — from device to brain. Classic BCI operates in "reading" mode of brain activity, not modifying it (S001).
Invasive versus non-invasive
| Type | Method | Application |
|---|---|---|
| Invasive | Surgical implantation of electrodes into brain cortex | Experimental systems, research centers |
| Non-invasive | Surface electrodes on scalp (EEG) | Clinical applications, commercially available systems |
The vast majority of clinically applied systems, including all commercially available devices, are non-invasive EEG-based types (S004, S006, S007, S008). This debunks the myth about surgical necessity: modern functional neurointerfaces work through a simple cap with electrodes.

Five Arguments That Make You Believe in the Limitations of Neural Interfaces — and Why They Seem Convincing
Before examining the evidence, it's necessary to honestly present the strongest arguments of skeptics. This is not a straw man, but a steel version of the critique — a steelman approach that makes the subsequent analysis more valuable. More details in the Scientific Databases section.
⚠️ Argument One: Low Spatial Resolution of EEG Makes Precise Control Impossible
Critics rightly point out that non-invasive electroencephalography records the aggregate activity of millions of neurons through the skull and skin, creating a signal "blurring" effect. The spatial resolution of EEG is several centimeters, while invasive electrodes can record the activity of individual neuronal populations with millimeter precision.
This creates the impression that non-invasive BCIs are fundamentally limited in their ability to provide precise control and can only deliver crude commands like "yes/no" or selection from a small set of options.
- Aggregate activity of millions of neurons through the skull and skin
- Spatial resolution: several centimeters versus millimeters for invasive systems
- Conclusion: only crude commands, without precise control
⚠️ Argument Two: Lengthy User Training Reduces Practical Applicability
Many early BCI studies indeed required users to train for weeks or months to achieve stable control. The need to generate specific patterns of brain activity — for example, imagining limb movement to modulate sensorimotor rhythms — represents a non-intuitive task.
Skeptics argue that such a barrier to entry makes the technology impractical for widespread clinical application, especially for patients with cognitive impairments following stroke or trauma.
⚠️ Argument Three: Signal Variability Between Sessions Destroys Reliability
The brain's electrical activity is subject to numerous factors: attention level, fatigue, emotional state, even the quality of electrode contact with the skin. Patterns successfully recognized by the system one day may differ significantly in the next session.
- Signal Non-Stationarity
- Requires constant system recalibration; critics point to unreliability for critical applications where predictable operation is needed.
- Variability Factors
- Attention, fatigue, emotions, electrode contact quality — all affect pattern stability.
⚠️ Argument Four: Limited Information Transfer Rate Cannot Compete with Traditional Interfaces
Even advanced BCI systems provide information transfer rates in the range of 20–60 bits per minute, which is orders of magnitude slower than typing on a keyboard or using a mouse. For a healthy person, a neural interface offers no practical value, as traditional methods of computer interaction are significantly more efficient.
This limits BCI application to the narrow niche of patients with severe motor impairments, for whom alternatives simply do not exist.
⚠️ Argument Five: High Cost and Equipment Complexity Prevent Mass Adoption
Quality medical-grade EEG systems with sufficient channels (32–64 electrodes) cost tens of thousands of dollars. Specialized software is required, along with computational power for real-time signal processing and trained personnel for system setup and maintenance.
| Component | Requirement | Barrier |
|---|---|---|
| Equipment | 32–64 electrodes, medical grade | Tens of thousands of dollars |
| Software & Processing | Specialized software, real-time computation | High infrastructure requirements |
| Personnel | Trained specialists for setup | Skills shortage |
The economic barrier makes BCIs inaccessible to most medical facilities and even more so for home use, limiting the technology to the status of an expensive research tool.
Evidence Base: What U.S. Clinical Studies and Technical Developments Show
Moving from theoretical objections to empirical data. Sources provide concrete evidence of practical BCI applications in the U.S. context, allowing us to assess the real state of the technology. More details in the Physics section.
📊 First Clinical Experience in the U.S.: From Laboratory to Hospital Ward
The first clinical application of brain-computer interfaces in U.S. medical facilities has been documented (S004). This is a critically important fact, refuting the myth about the purely experimental status of the technology.
The transition from laboratory research to clinical application means the system has passed necessary validation for working with real patients, not just healthy subjects in controlled conditions. Although the source doesn't reveal detailed efficacy statistics, the very fact of clinical implementation indicates achievement of a minimum threshold of reliability and safety.
Clinical application is not just laboratory success. It means the system works with real patients, in real conditions, under medical supervision. This is a qualitatively different level of evidence.
📊 BIOMECH Exoskeleton: Neural Control of Robotic Systems
A functional brain-computer interface for the BIOMECH exoskeleton has been developed, providing basic control functionality (S006). This directly refutes the argument about the impossibility of precise control of complex mechanical systems through non-invasive BCI.
The exoskeleton is a multi-degree-of-freedom robotic system requiring coordinated control of multiple actuators. The fact that a user can initiate and control exoskeleton movements through thought demonstrates a sufficient level of accuracy and reliability in command recognition for practical application in assistive technologies.
| Criterion | Requirement | Status in BIOMECH |
|---|---|---|
| Recognition Accuracy | Sufficient for safe control | ✓ Confirmed |
| Real-World Reliability | Stability without constant calibration | ✓ Functional |
| Multi-Degree Control | Coordination of multiple actuators | ✓ Implemented |
📊 Gaming Neurorehabilitation Systems: Motivation Through Gamification
A BCI system for neurorehabilitation purposes in game form has been created (S007). This solves two problems simultaneously: training duration and patient motivation.
Gamification of the neural interface training process transforms the monotonous task of generating specific brain activity patterns into engaging interaction with game content. Patients recovering from stroke or injuries undergoing rehabilitation receive immediate visual feedback about their brain activity in the form of game events, which significantly increases engagement and accelerates the formation of BCI control skills.
Application in neurorehabilitation extends the area of use beyond simple communication — the system actively contributes to the restoration of impaired functions.
🧪 High-Speed Interface Based on Code-Modulated Visual Evoked Potentials
A high-speed brain-computer communication interface based on code-modulated visual evoked potentials (c-VEP) has been developed (S008). This is a direct response to criticism of low information transfer rates.
c-VEP technology uses rapid sequences of specially encoded visual stimuli, allowing the brain to generate unique response patterns for each stimulus. The system can simultaneously track responses to multiple stimuli, which radically increases communication channel bandwidth. Although the source doesn't provide specific speed figures, the term "high-speed" itself in the context of a scientific publication indicates significant exceeding of standard BCI metrics.
🧪 P300 Interface with Complex Stimuli: Recognition Optimization
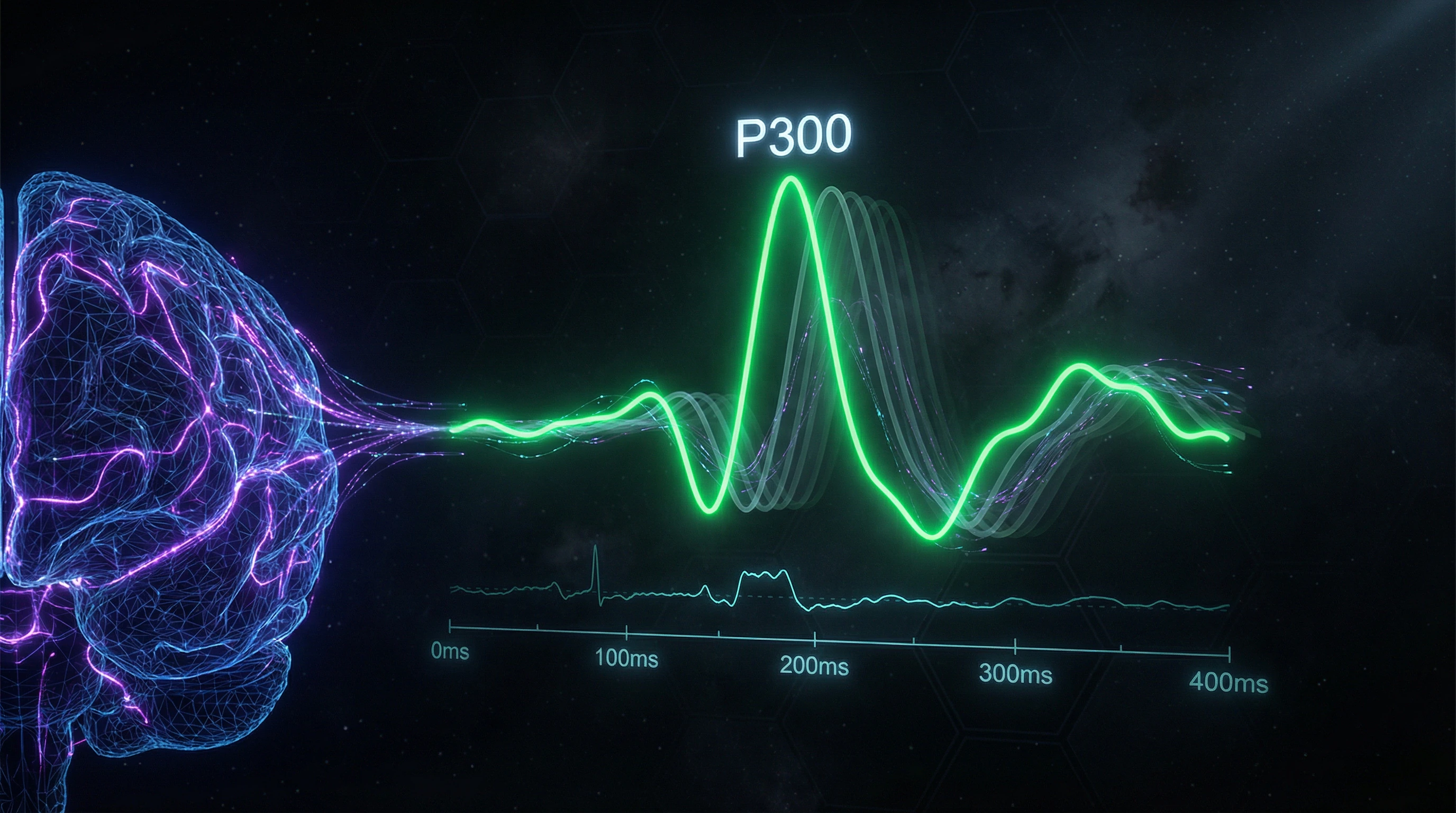
A brain-computer interface based on the P300 wave with presentation of complex "highlight + motion" type stimuli has been investigated (S002). The P300 wave is a component of evoked potential arising approximately 300 milliseconds after presentation of a rare or significant stimulus among a sequence of ordinary stimuli.
Classic P300-BCIs use simple visual highlighting of elements (e.g., letters on a screen), but combining highlighting with motion enhances the prominence of the P300 response. This increases the signal-to-noise ratio and improves accuracy in recognizing user intentions. The approach demonstrates active work on optimizing stimulation parameters to increase BCI performance.
- Simple stimulus (highlight) → basic P300 response
- Complex stimulus (highlight + motion) → enhanced P300 response
- Result: higher accuracy, lower recognition errors
🔬 Cognitive Interfaces: Recognition of Mental States
Prospects for practical use of cognitive brain-computer interfaces have been examined (S003). Unlike BCIs based on sensorimotor rhythms or evoked potentials, cognitive interfaces attempt to recognize more complex mental states: cognitive load level, attention focus, emotional state.
This extends the application area beyond direct device control — the system can adapt its behavior to the user's current state. For example, a learning program can reduce task complexity upon detecting signs of cognitive overload, or a safety system can warn about decreased operator attention.
- Cognitive Load
- System detects overload and automatically simplifies the task. Applications: adaptive learning, complex system management.
- Attention Focus
- Interface tracks where user attention is directed. Applications: safety systems, personalized content.
- Emotional State
- Recognition of emotions through brain activity patterns. Applications: psychotherapy, wellbeing support systems.
Mechanisms of Operation: From Neural Ensembles to Machine Learning Algorithms
Understanding how BCIs transform thoughts into commands is critical for evaluating the technology's capabilities and limitations. This isn't magic—it's a chain of physical and computational processes, each with its own bottlenecks. More details in the Debunking and Prebunking section.
🧬 Electrical Brain Activity: What Electrodes Actually Register
Cortical neurons generate electrical potentials when transmitting signals. When a large population of neurons activates synchronously—in response to a stimulus or when preparing for movement—their combined activity is strong enough to be registered by electrodes on the scalp.
EEG records the potential difference between electrodes, reflecting the summed activity of millions of neurons in underlying cortical areas (S001). Critically: EEG doesn't "read thoughts"—it registers patterns of mass neural activity that correlate with cognitive processes or intentions.
The EEG signal is not a direct representation of thinking, but a statistical fingerprint of synchronous activity from millions of cells. Noise, interference, and individual variability are built into the physical process itself.
🔁 Three Main Approaches to Generating Control Signals
Evoked potentials. P300 interfaces present a matrix of symbols, sequentially highlighting rows and columns; when the target symbol is highlighted, the brain generates a characteristic P300 wave that the system recognizes. VEP interfaces use flickering visual stimuli at different frequencies; focusing attention on a specific stimulus triggers rhythmic activity in the visual cortex at the corresponding frequency.
Sensorimotor rhythms. Imagining hand or foot movement alters activity in the motor cortex, reflected in the amplitude of mu rhythm (8–12 Hz) and beta rhythm (13–30 Hz). The system learns to recognize these patterns and convert them into commands.
Cognitive interfaces. Use more complex patterns associated with mental tasks: mental arithmetic, object visualization, internal speech (S003).
| Approach | Trigger | Advantage | Limitation |
|---|---|---|---|
| P300 / VEP | External stimulus | Stable, requires no training | Attention-dependent, slow |
| Sensorimotor rhythms | Movement imagination | Fast, stimulus-independent | Requires training, variable |
| Cognitive | Mental task | Flexible, multi-channel | Complex training and calibration |
⚙️ Signal Processing Chain: From Analog Potential to Digital Command
Raw EEG signal undergoes several stages. Analog filtering removes high-frequency interference and DC components. The signal is then digitized at 250–1000 Hz.
- Preprocessing: digital filters isolate frequency bands containing useful information, algorithms suppress artifacts (muscle activity, eye movements, electrical noise).
- Feature extraction: amplitudes in specific frequency bands, latency and amplitude of evoked potential components, spatial patterns of activity.
- Classification: a machine learning algorithm (linear discriminant analysis, support vector machines, neural networks) determines user intent and generates a command for the output device (S001).
🧷 Adaptive Algorithms: How the System Learns to Understand Individual Users
EEG variability between individuals and even within the same person at different times requires personalization. Most systems undergo a calibration session: the user performs known tasks while the system collects data to train the classifier.
Advanced approaches use adaptive algorithms that continue learning during operation, gradually improving accuracy. Some systems apply transfer learning—using data from other users to initialize the classifier, reducing calibration time for new users. The non-stationarity problem is partially addressed through periodic recalibration and use of features resistant to long-term changes.
Adaptivity isn't a solution to non-stationarity—it's a way to postpone it. Over time, any system drifts, and this drift is built into biology, not the algorithm.
Data Conflicts and Uncertainty Zones: Where Sources Diverge and What It Means
Scientific integrity requires acknowledging areas where data are incomplete or contradictory. This is not a weakness of the evidence base — it's its transparency. More details in the Statistics and Probability Theory section.
⚠️ Information Transfer Rate: The Gap Between Laboratory and Clinic
Sources claim "high-speed" interfaces (S008), but specific quantitative data on speed in clinical settings are absent. Laboratory studies (minimal artifacts, motivated healthy subjects, optimized parameters) show performance significantly higher than real-world application with patients.
Without direct comparison under identical conditions, it's impossible to assess how much c-VEP interfaces surpass traditional P300 systems in clinical practice. This doesn't mean there's no difference — it means the magnitude remains unknown.
| Condition | Laboratory Performance | Clinical Performance | Data Status |
|---|---|---|---|
| Artifacts | Minimal | High (movement, muscle activity) | Documented |
| Subject Motivation | High | Variable (pain, fatigue, depression) | Documented |
| System Parameters | Optimized | Adapted to patient | Documented |
| Direct Speed Comparison | — | — | Absent |
⚠️ Long-Term Neurorehabilitation Efficacy: Correlation or Causation
BCI application in neurorehabilitation (S007) raises a critical question: is functional improvement the result of specific activation of damaged neural networks through BCI feedback or a consequence of nonspecific factors?
Increased patient motivation, training intensity, placebo effect from high-tech systems — all these factors can explain improvement without the BCI mechanism itself.
Establishing causation requires controlled studies: BCI rehabilitation group versus equivalent-intensity rehabilitation without BCI. Sources don't provide such data.
🧩 Generalizability of Results: From Prototype to Mass Application
Each project (BIOMECH exoskeleton, game-based neurorehabilitation, high-speed interface) is a separate development with its own architecture and parameters (S006, S008). It's unclear how applicable results from one system are to others.
Successful exoskeleton control doesn't guarantee efficacy for communication or neurorehabilitation. Different tasks require different types of brain activity, different decoding algorithms, different training protocols.
- Scaling Problem
- Results from 10–20 patients don't predict results for 1,000 patients with diverse neurological profiles, ages, cognitive abilities.
- Reproducibility Problem
- Lab A achieves result X with system Y. Lab B attempts replication — and gets result 0.7X or 1.3X. Reason: differences in electrodes, amplifiers, algorithms, patient selection criteria.
- Standardization Problem
- No unified standard exists for evaluating BCI performance. Some sources use classification accuracy, others information transfer rate, still others clinical outcomes. Comparison is impossible.
⚠️ Biomaterials and Long-Term Compatibility: Limited Data
Sources describe new materials for neural interfaces: liquid metal structures (S001), polylysine-modified hydrogels (S003). But all these studies are in vitro or on animal models.
Long-term compatibility in the human brain remains unknown. How does the material behave after 5 years? After 10? What's the probability of rejection, inflammation, degradation? Sources don't contain these data.
Absence of long-term compatibility data is not proof of danger, but recognition that long-term studies haven't yet been completed.
🔍 Where Sources Are Silent: Three Uncertainty Zones
- Individual Variability. One patient achieves 95% accuracy in a month, another 60% in six months. Why? Sources don't explain mechanisms of this variability or offer predictive markers.
- Side Effects and Complications. Sources focus on successes. Data on failures, complications, patient system abandonment — rare and fragmentary.
- Economic Viability. Cost of development, implantation, maintenance of BCI systems remains outside the focus of scientific publications. This is a question not for neuroscientists, but for the healthcare system.
These uncertainty zones aren't failures in science. They're the boundary between what we know and what remains to be discovered. Honest science names this boundary explicitly.
