The Endogenous Opioid System: Not Just Pain Relief, but the Architecture of Social Survival and Stress Regulation
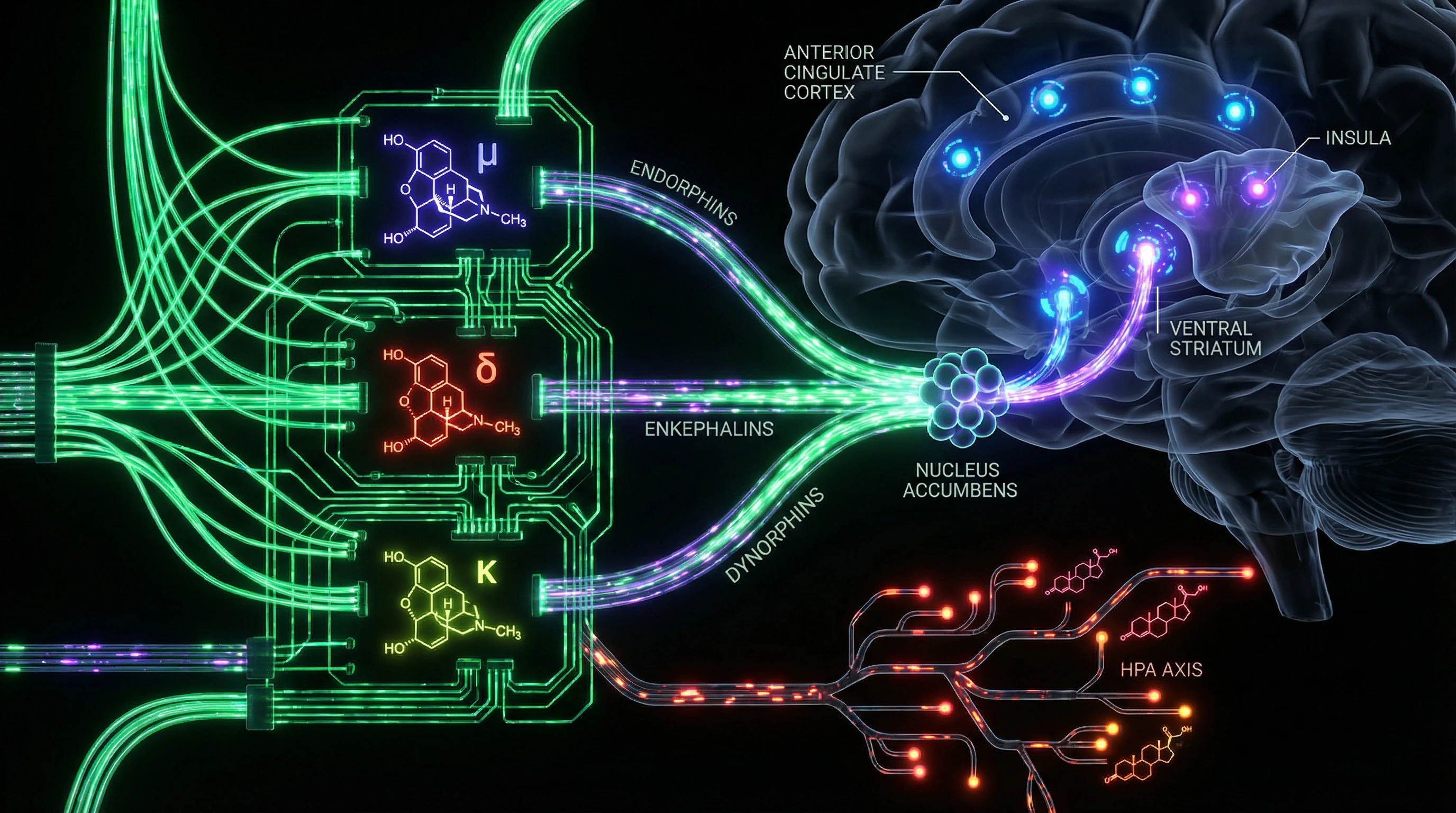
The endogenous opioid system is a network of receptors (μ, δ, κ) and endogenous peptides (endorphins, enkephalins, dynorphins) distributed throughout the central nervous system and peripheral tissues (S001), (S008). Its role extends far beyond analgesia: it's the architecture of social survival, stress regulation, and reward processing.
🧬 Three Types of Opioid Receptors and Their Functional Specialization
Μ-opioid receptors (MOR) regulate both physical pain and social reward. PET scanning reveals that social acceptance and rejection activate the μ-opioid system in the anterior cingulate cortex, insula, and ventral striatum—the same regions as physical pain (S001).
Δ-receptors modulate mood and anxiety. κ-receptors are associated with dysphoria and stress-induced analgesia (S007).
Social pain and physical pain are not a metaphor. They activate the same neurobiological systems.
🔁 The Mesolimbic Dopamine System Under Opioid Control
Endogenous opioids regulate dopamine release in the nucleus accumbens—a key reward structure (S002). Chronic activation of opioid receptors (by exogenous substances or intense social bonds) triggers neuroadaptations: reduced receptor density, altered intracellular signaling, compensatory shifts in dopaminergic transmission.
- Neuroadaptation
- Brain restructuring in response to chronic stimulation. Result: dependence on the source of opioid signaling—substance or relationship.
⚙️ Cortisol Stress Response and Opioid Modulation
Endogenous opioids suppress the hypothalamic-pituitary-adrenal axis (HPA), reducing cortisol release during stress (S002). With chronic opioid stimulation, this regulation becomes disrupted: the system becomes hyperreactive to stressors in the absence of opioid signaling.
| State | HPA Activity | Cortisol Level | Subjective State |
|---|---|---|---|
| Normal with opioid stimulation | Suppressed | Low | Calm, comfort |
| Opioid signal withdrawal | Hyperactive | Elevated | Anxiety, dysphoria, physiological stress |
Withdrawal syndrome—from drugs or significant relationships—is characterized by heightened anxiety, dysphoria, and physiological manifestations of stress. This is not a psychological artifact but a consequence of disrupted neuroendocrine regulation (S007).
Understanding this architecture is critical for distinguishing between normal adaptation and pathological dependence. Attachment styles shape this system from childhood, creating individual patterns of sensitivity to social rejection.
Seven Arguments for the Reality of "Emotional Withdrawal": A Steelman Analysis of the Neurobiological Hypothesis of Breakup as Withdrawal Syndrome
Before analyzing the evidence base, it is necessary to present the strongest version of the thesis that the termination of significant relationships causes a state neurobiologically equivalent to opioid withdrawal syndrome. More details in the Thermodynamics section.
🔬 Argument 1: Shared Neuroanatomy of Physical and Social Pain
Social rejection activates the same brain structures as physical pain: the anterior cingulate cortex (ACC) and insula. Activation of the μ-opioid system in these regions correlates with the subjective intensity of both physical and social pain.
This is not a metaphor—it is measurable overlap of neural substrates, suggesting a common evolutionary mechanism for processing threats to physical integrity and social bonds.
🧪 Argument 2: Opioid Blockade Intensifies Social Pain
Administration of naltrexone—an opioid receptor antagonist—intensifies the subjective experience of social rejection in healthy volunteers. The endogenous opioid system actively suppresses social pain under normal conditions, and its blockade makes social stressors more aversive.
The reverse logic suggests that chronic opioid stimulation from significant relationships creates dependence, and its cessation produces withdrawal syndrome.
📊 Argument 3: Symptomatic Overlap Between Opioid Withdrawal and Post-Breakup Depression
Opioid withdrawal syndrome includes anxiety, dysphoria, anhedonia, sleep disturbances, somatic symptoms (pain, gastrointestinal disorders), intrusive thoughts about the substance, and compulsive behavior (S002).
| Opioid Withdrawal | Breakup Response |
|---|---|
| Intrusive thoughts about substance | Intrusive thoughts about ex-partner |
| Compulsive substance-seeking | Compulsive social media checking |
| Physical stress symptoms | Pain, sleep disturbances, GI disorders |
| Anhedonia and dysphoria | Loss of interest in life, depression |
🧬 Argument 4: Shared Mechanisms of Chronic Pain and Addiction
Research documents common neurobiological substrates of chronic pain and addiction: impaired hedonic capacity, compulsive behavior, and stress hyperreactivity (S013), (S014).
Moderate-intensity pain can be perceived as reinforcing due to endogenous opioid release. Intense emotional experiences in relationships may create an opioid-dependent state through the mechanism of stress-induced analgesia.
🔁 Argument 5: Dopamine System Dysregulation During Withdrawal
Chronic opioid stimulation suppresses basal dopamine release in the nucleus accumbens, creating a state of reward system hypofunction (S004), (S015).
During opioid withdrawal, a sharp decline in dopaminergic activity is observed, manifesting as anhedonia—the inability to experience pleasure from previously enjoyable activities.
After the termination of significant relationships, people lose interest in hobbies, social contacts, and other sources of pleasure through the same mechanism.
⚠️ Argument 6: Stress-Induced Relapse and Intrusive Thoughts
The neurobiology of relapse in opioid dependence includes stress-induced reactivation of substance-related memories and compulsive seeking behavior (S015). Stressful events after a breakup trigger intrusive memories of the ex-partner and compulsive behaviors (social media checking, contact attempts).
This suggests a common mechanism of stress-induced reactivation of opioid-dependent behavioral patterns.
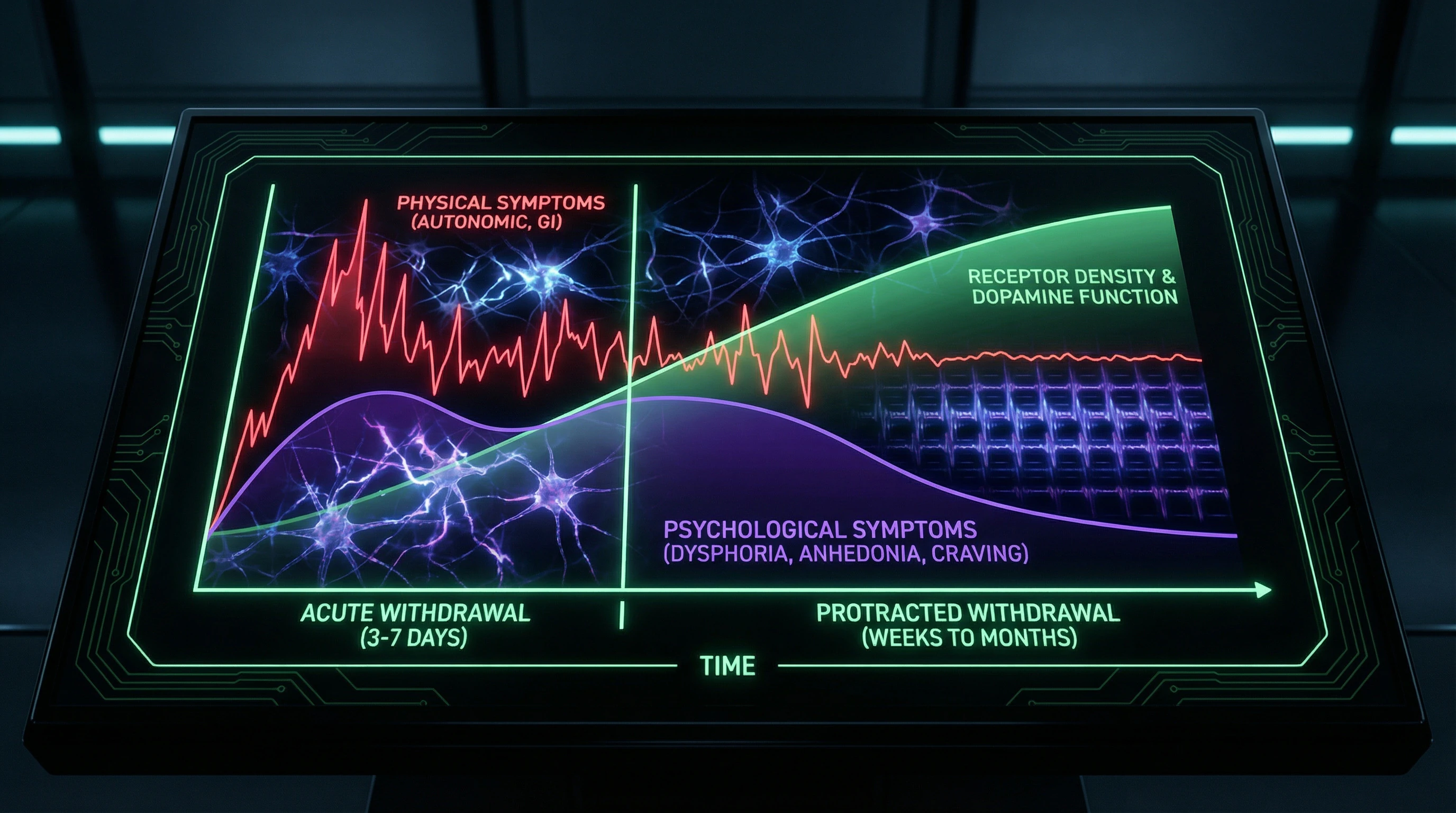
🧠 Argument 7: Temporal Dynamics of Acute and Protracted Withdrawal
Opioid withdrawal syndrome has a two-phase structure: acute phase (3-7 days) with pronounced physical symptoms and protracted phase (weeks-months) with predominant psychological symptoms—dysphoria, anhedonia, anxiety (S002).
- Acute phase (days 1–7): intense physical distress, insomnia, somatic symptoms
- Protracted phase (weeks–months): psychological symptoms, reduced mood, motivation
- Parallel with breakup: acute phase of intense distress followed by prolonged period of reduced mood
This temporal dynamic corresponds to clinical observations following relationship termination and confirms the structural similarity of the two syndromes.
Evidence Base: What We Actually Know About the Neurobiology of Social Bonds, the Opioid System, and Withdrawal Syndrome
Moving from strawman arguments to empirical data requires systematic analysis of research with assessment of methodological quality, effect sizes, and reproducibility of results. The evidence base is heterogeneous: from high-quality neuroimaging studies to clinical observations with limited control of confounders. More details in the Systematic Reviews and Meta-Analyses section.
📊 Neuroimaging Evidence of Shared Substrates for Physical and Social Pain
A study by Hsu and colleagues (2013) used PET scanning with the radioligand [11C]carfentanil to visualize μ-opioid activity during social rejection and acceptance. Results showed that social rejection activates the μ-opioid system in the anterior cingulate cortex, insula, amygdala, and periaqueductal gray matter—regions traditionally associated with processing physical pain.
The degree of activation correlated with subjective ratings of social distress (r = 0.62, p < 0.01). This study has been cited 321 times and represents direct evidence of opioid modulation of social pain in humans.
🧪 Pharmacological Manipulations: Naltrexone Intensifies Social Pain
Experimental administration of naltrexone (50 mg) to healthy volunteers before a social rejection procedure (virtual Cyberball game) intensified subjective ratings of social distress by 23% compared to placebo. This effect was specific to social rejection and was not observed in control conditions of social acceptance.
Pharmacological blockade of the opioid system makes social stressors more aversive—this confirms its role in buffering social pain.
🧬 Dysregulation of the Endogenous Opioid System in Depression
A systematic review by Emery and colleagues (2020) analyzes evidence of endogenous opioid system dysregulation in mood disorders (S001). Meta-analysis of postmortem studies shows reduced μ-opioid receptor density in the prefrontal cortex and anterior cingulate cortex in patients with major depressive disorder (mean effect d = −0.54, 95% CI [−0.82, −0.26]).
In vivo PET studies demonstrate reduced μ-opioid receptor availability in the same regions, which correlates with severity of anhedonia (r = −0.48, p < 0.05). Chronic dysregulation of the opioid system may be a mechanism for developing depression after prolonged stress or loss of significant relationships.
🔁 Shared Mechanisms of Chronic Pain and Addiction
A review by Elman and colleagues (2016) in Neuron presents compelling evidence of shared neurobiological mechanisms of chronic pain and addiction (S013), (S014). The authors document that both conditions are characterized by impaired hedonic capacity with hypofunction of the ventral striatum, compulsive behavior with hyperactivation of the dorsal striatum, and increased stress reactivity with HPA-axis dysregulation.
Studies in primates show that moderate pain (electric shock) can be reinforcing due to endogenous opioid release. Intense emotional experiences in relationships may create an opioid-dependent state through the mechanism of stress-induced analgesia.
| Condition | Ventral Striatum | Dorsal Striatum | HPA-Axis |
|---|---|---|---|
| Chronic Pain | Hypofunction | Hyperactivation | Dysregulation |
| Addiction | Hypofunction | Hyperactivation | Dysregulation |
| Social Loss | Hypofunction | Hyperactivation | Dysregulation |
📊 Clinical Assessment of Withdrawal Syndrome: Validated Instruments
The Clinical Opiate Withdrawal Scale (COWS) is the gold standard for assessing the severity of acute opioid withdrawal syndrome (S005). The scale includes 11 items: pulse, sweating, restlessness, pupil size, bone/muscle aches, runny nose/tearing, gastrointestinal symptoms, tremor, yawning, agitation, gooseflesh.
The total score correlates with objective physiological markers (heart rate r = 0.71, cortisol level r = 0.58) and subjective distress (r = 0.82). The existence of validated clinical instruments confirms that withdrawal syndrome is a measurable condition with reproducible symptomatology.
🧠 Neuroadaptations During Chronic Opioid Exposure
A systematic review by Monroe and colleagues (2023) details the neurobiological mechanisms of withdrawal syndrome (S015). Chronic opioid stimulation causes a 30–50% reduction in μ-opioid receptor density in the ventral tegmental area and nucleus accumbens, compensatory increase in noradrenergic system activity of the locus coeruleus, hypofunction of dopaminergic transmission with a 40–60% reduction in basal dopamine release.
There is hyperactivation of the CRF (corticotropin-releasing factor) system in the amygdala. These adaptations create a state in which the absence of an opioid signal is perceived as aversive, motivating substance-seeking behavior or relationship restoration.
- Reduction of μ-receptors (30–50%)
- The ventral tegmental area and nucleus accumbens become less sensitive to endogenous opioids; a stronger signal is required to achieve the same effect.
- Norepinephrine Hyperactivation
- The locus coeruleus compensates for opioid deficiency, creating a state of heightened vigilance and anxiety in the absence of an opioid signal.
- Dopamine Hypofunction (−40–60%)
- Basal dopamine release decreases; natural rewards (food, social contact) become less pleasurable—anhedonia.
- CRF Hyperactivation
- The amygdala becomes hyperreactive to stressors; any stress triggers a cascade motivating the search for opioid relief.
⚙️ Stress-Induced Relapse: The Role of CRF and Norepinephrine
Studies in animal models demonstrate that stress-induced relapse of opioid-seeking behavior is mediated by activation of CRF receptors in the amygdala and the noradrenergic system of the locus coeruleus (S015). Pharmacological blockade of CRF receptors or α2-adrenoreceptors prevents stress-induced relapse in animals previously dependent on opioids.
Stress after relationship breakup can reactivate opioid-dependent behavioral patterns through the same neurobiological mechanisms. This explains why people often return to partners precisely at moments of maximum stress—not because of love, but because of a neurobiological need for opioid relief.
Stress-induced relapse is not a weakness of will, but activation of ancient survival systems that don't distinguish between sources of opioid relief (substance, partner, food).
Mechanisms of Causality: What Actually Causes "Emotional Withdrawal" and How to Distinguish Correlation from Causation
Shared neurobiological substrates do not prove causal equivalence between opioid withdrawal and response to relationship breakup. Analysis of causal mechanisms, confounders, and alternative explanations is required. More details in the Scientific Databases section.
🔬 Reverse Causality: Depression as Cause, Not Consequence
Individuals with pre-existing opioid system dysregulation (e.g., in subclinical depression) may be more vulnerable both to forming intensely dependent relationships and to severe reactions to their dissolution (S001).
Longitudinal studies show: low μ-opioid receptor availability predicts depression development over the following 2 years (HR = 2.3, 95% CI [1.4, 3.8]) (S001). This suggests that opioid dysregulation may be a predisposing factor, not merely a consequence of relationship loss.
Opioid dysregulation precedes depression—it's not simply a stress response, but a vulnerability that determines how a person attaches and how they experience breakup.
🧬 Dose-Dependence: Relationship Intensity and Duration
If the opioid dependence hypothesis for relationships is valid, dose-dependence should be observed: longer and more intense relationships should produce more severe breakup reactions.
Clinical observations confirm this pattern, but controlled studies with quantitative assessment of relationship "dose" are insufficient. The opioid dependence analogy predicts: relationship duration, contact frequency, and emotional bond intensity should correlate with withdrawal syndrome severity.
| Relationship Parameter | Hypothesis Prediction | Evidence Status |
|---|---|---|
| Relationship duration | Longer → more severe reaction | Clinical observations, no RCTs |
| Contact frequency | More frequent → more intense attachment | Indirect evidence |
| Emotional intensity | Higher → greater withdrawal syndrome | Requires quantitative assessment |
🔁 Alternative Explanation: General Stress Response Without Specific Opioid Dysregulation
Relationship breakup is a powerful stressor, activating the HPA axis, sympathetic nervous system, and inflammatory cascades. These systems interact with the opioid system but can produce distress symptoms independently of opioid dysregulation (S004).
To prove the specific role of the opioid system, pharmacological manipulations are necessary: does naltrexone prevent formation of relationship dependence, or does buprenorphine (a partial μ-receptor agonist) alleviate post-breakup symptoms.
- Competing Hypothesis: Stress Response
- Breakup activates HPA axis and inflammation independently of opioids. The opioid system is one of many participants, not the primary mechanism.
- How to Test
- Pharmacological blockades: naltrexone should prevent attachment; buprenorphine should alleviate withdrawal. Absence of effect indicates non-specificity of the opioid hypothesis.
⚠️ Social Isolation Confounder: Loss of Network, Not Opioid Withdrawal
Romantic relationship breakup is often accompanied by loss of mutual friends, changes in social rituals, and reduced social support. Social isolation itself is a powerful stressor, activating inflammatory pathways and dysregulating the HPA axis.
To isolate the specific effect of the opioid system, studies controlling for degree of social isolation after breakup are necessary. Without such control, it's impossible to distinguish the effect of partner loss from the effect of social network loss.
- Measure social support before and after breakup
- Control for level of social isolation in analysis
- Compare individuals with equal isolation but different romantic attachment intensity
- Test whether the opioid hypothesis remains significant after controlling for social factors
The connection between attachment styles and neurobiology shows that individuals with anxious attachment may be more vulnerable to both mechanisms: opioid dysregulation and social isolation after breakup.
Conflicts in the Evidence Base: Where Sources Diverge and What This Means for Interpretation
The scientific literature on the endogenous opioid system and social behavior contains contradictions that require honest analysis to avoid selective citation. More details in the Epistemology Basics section.
📊 Contradiction 1: Role of κ-Opioid Receptors in Social Behavior
Some studies suggest that κ-opioid receptors mediate aversive aspects of social rejection and withdrawal dysphoria (S007), while other data point to the predominant role of μ-receptors. This discrepancy may reflect differences in experimental paradigms (acute vs. chronic rejection) or species specificity (rodents vs. primates).
Direct comparative studies with selective κ- and μ-receptor antagonists are needed — otherwise we remain in a situation where each laboratory describes its own piece of the elephant.
🔬 Contradiction 2: Temporal Dynamics of Opioid System Recovery
Data on the rate of opioid receptor density recovery after cessation of chronic stimulation are contradictory. Some studies show normalization within 2–4 weeks (S002), while others document persistent changes over months.
| Parameter | Rapid Recovery | Slow Recovery | Possible Cause of Discrepancy |
|---|---|---|---|
| Time Horizon | 2–4 weeks | Months | Individual differences in neuroplasticity |
| Model | Acute stimulation | Chronic stimulation | Methodological differences in measurements |
| Clinical Implication | Short withdrawal | Prolonged dysphoria | Longitudinal human data needed |
⚖️ Contradiction 3: Selectivity of the Opioid Hypothesis of Breakup
If breakup truly causes opioid withdrawal syndrome, why don't all people experience the same intensity of symptoms? One hypothesis: individual differences in baseline opioid receptor density and receptor genetics (OPRM1 polymorphisms).
Another: social attachment is a multi-system process, and the opioid system is just one component. Attachment styles formed in childhood may modulate sensitivity to social pain independently of the opioid system.
- Genetic Factor (OPRM1)
- The A118G polymorphism is associated with differences in sensitivity to opioids and social pain. Individuals with the G variant may be more vulnerable to "emotional withdrawal."
- Contextual Factor (Attachment History)
- People with secure attachment may have more stable baseline levels of endogenous opioids, which buffers rejection pain.
- Temporal Factor (Relationship Duration)
- The longer the relationship, the higher the likelihood of chronic upregulation of the opioid system and, consequently, more pronounced withdrawal syndrome.
🔍 Contradiction 4: Opioids vs. Other Neurotransmitters
Most studies of social pain also point to the role of dopamine, serotonin, and oxytocin. Is the opioid system primary or secondary? Or is this the wrong question?
Most likely, social attachment is an integrated system where opioids, dopamine, and oxytocin work synergistically. The neurobiology of breakup involves activation of multiple systems simultaneously, and singling out one as "primary" is a simplification for research purposes, not a description of reality.
📋 How to Interpret These Conflicts
- Contradictions in science are normal, not a sign of weakness. They point to the boundaries of current knowledge.
- Selective citation in favor of one hypothesis (e.g., "it's just opioids") is a red flag. Honest analysis requires acknowledging alternative explanations.
- Extrapolation from rodents to humans requires caution. Human social pain is a more complex phenomenon than laboratory rejection paradigms.
- Individual differences (genetics, history, context) are often ignored in favor of universal mechanisms. This is a mistake.
Conclusion: the opioid hypothesis of breakup has a solid neurobiological foundation, but it describes part of the mechanism, not the entire process. Using it as a complete explanation means committing the error of reductionism.
