 🧠 Neuroscience
🧠 NeuroscienceNeuroscience: Exploring the Brain and Nervous Systemλ
An interdisciplinary science studying the structure, function, and development of the nervous system, from molecular mechanisms to human behavior and cognition.
Overview
Neuroscience integrates biology, physics, chemistry, and computer science to decode 🧠 how the brain works — from molecular mechanisms to consciousness. Modern technologies (fMRI, PET scanning) allow us to observe neural network activity in real time and understand how memory, emotions, and decisions are formed. The findings are applied in medicine, education, AI development, and cognitive therapy.
🛡️
Laplace Protocol: Neuroscience is grounded in rigorous experimental methods and reproducible results, separating scientifically validated knowledge about the brain from popular myths and pseudoscientific claims about how it functions.
Reference Protocol
Scientific Foundation
Evidence-based framework for critical analysis
Protocol: Evaluation
Test Yourself
Quizzes on this topic coming soon
Sector L1
Articles
Research materials, essays, and deep dives into critical thinking mechanisms.
 🧠 Neuroscience
🧠 Neuroscience 🧠 Neuroscience
🧠 Neuroscience 🧠 Neuroscience
🧠 Neuroscience 🧠 Neuroscience
🧠 Neuroscience 🧠 Neuroscience
🧠 Neuroscience 🧠 Neuroscience
🧠 Neuroscience 🧠 Neuroscience
🧠 Neuroscience 🧠 Neuroscience
🧠 Neuroscience 🧠 Neuroscience
🧠 Neuroscience 🧠 Neuroscience
🧠 Neuroscience 🧠 Neuroscience
🧠 Neuroscience 🧠 Neuroscience
🧠 Neuroscience⚡
Deep Dive
Neural System Architecture: From Molecular Machines to Global Networks
The nervous system is a hierarchical structure where billions of specialized cells coordinate all bodily processes from automatic reflexes to abstract thinking. Neurons transmit information across distances from micrometers to meters at speeds up to 120 m/s through unique morphology and electrochemical properties.
The human brain contains approximately 86 billion neurons, each forming an average of 7,000 synaptic contacts. This network creates the foundation for all cognitive processes.
Neurons and Synaptic Transmission: Molecular Logic of Communication
A neuron consists of the soma (cell body), dendrites (signal reception), and axon (transmission). Information is encoded by action potentials—stereotypical electrical impulses with amplitude around 100 mV and duration of 1–2 ms, generated when membrane depolarization exceeds the threshold of −55 mV.
Action potential generation is enabled by cascading opening of voltage-gated sodium channels, passing up to 10⁷ Na⁺ ions per second through a single channel. The myelin sheath increases conduction velocity 5–50 fold through saltatory conduction—jumping propagation of excitation between nodes of Ranvier.
Synaptic transmission occurs predominantly through chemical means via neurotransmitter release into the synaptic cleft, 20–40 nm wide. Action potential arrival at the presynaptic terminal opens voltage-gated calcium channels; Ca²⁺ influx triggers synaptic vesicle exocytosis within 0.2–0.5 ms.
- Neurotransmitter Quantum
- Contents of a single synaptic vesicle; a typical synapse releases 1–5 quanta per impulse with probability 0.1–0.9.
- Glutamate
- Primary excitatory neurotransmitter (80–90% of CNS synapses); its balance with GABA determines neural network excitability.
- GABA
- Primary inhibitory neurotransmitter (10–20% of synapses); critical for preventing hyperexcitability.
Central and Peripheral Nervous Systems: Functional Division of Labor
The CNS includes the brain (mass 1300–1400 g) and spinal cord (length 40–45 cm, 31 segments), protected by bony structures and three meningeal layers. The cerebral cortex contains 16–20 billion neurons organized in six layers 1.5–4.5 mm thick; total cortical surface area when flattened is 1800–2300 cm².
| Structure | Function | Mechanism |
|---|---|---|
| Basal Ganglia | Movement Control | Selection and initiation of motor programs |
| Hippocampus | Memory Consolidation | Encoding episodic events |
| Amygdala | Emotional Responses | Threat and reward assessment |
| Spinal Cord | Reflexes and Motor Function | 30–50 ms latency through 31 pairs of nerves |
The PNS consists of 12 pairs of cranial and 31 pairs of spinal nerves, divided into the somatic system (voluntary control of skeletal muscles) and autonomic system (involuntary regulation of internal organs).
- Sympathetic Division (norepinephrine): fight-or-flight response—increases heart rate from 60–80 to 120–180 bpm, redistributes blood flow to muscles (up to 80% of cardiac output), mobilizes energy reserves.
- Parasympathetic Division (acetylcholine): rest-and-digest mode—stimulates digestion, reduces heart rate, activates anabolic processes.
- Enteric Nervous System: 200–600 million neurons in the gut function autonomously and bidirectionally interact with the CNS via the vagus nerve.
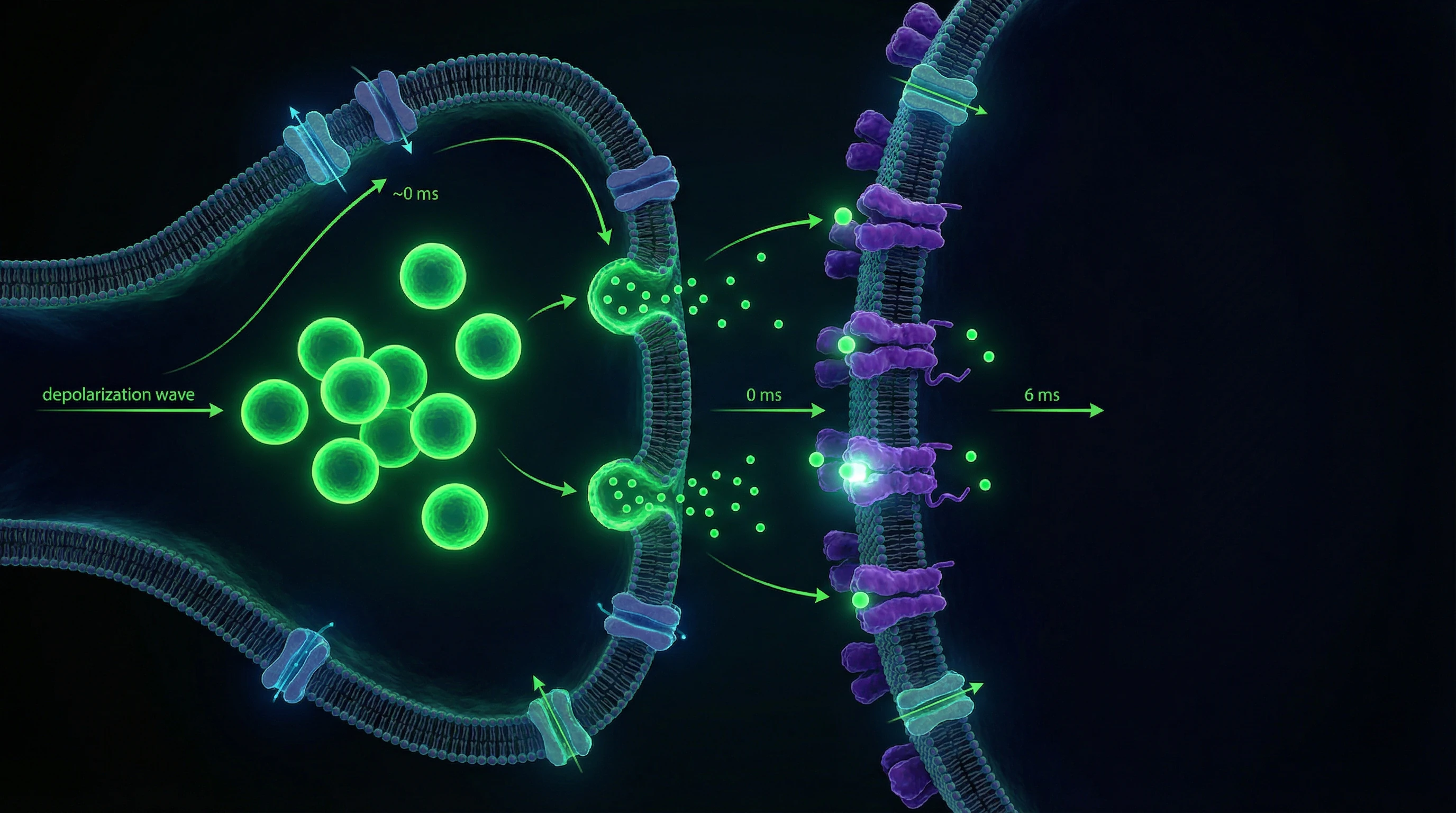
Brain Research Methods: From Electrodes to Molecular Markers
Modern neuroscience uses a multimodal approach, combining methods with varying spatial (from nanometers to centimeters) and temporal (from microseconds to years) resolution. Breakthroughs of recent decades — functional MRI (1990s), optogenetics (2005), two-photon in vivo microscopy (2000s), connectomics (2010s) — have enabled the transition from correlational observations to causal analysis of neural mechanisms.
Integration of data across different scales through computational modeling creates multilevel brain maps from molecules to behavior.
Neuroimaging and Electrophysiology: Observing the Living Brain
Functional MRI (fMRI) detects blood flow changes through the BOLD signal (blood oxygen level dependent) with spatial resolution of 1–3 mm and temporal resolution of 1–2 s, allowing noninvasive mapping of whole-brain activity. Neuronal activation increases local blood flow by 20–40% with a 4–6 s delay, exceeding oxygen demand (neurovascular coupling), creating contrast between active and inactive zones.
Positron emission tomography (PET) uses radioactive tracers to visualize glucose metabolism (¹⁸F-FDG), neurotransmitter systems (¹¹C-raclopride for dopamine), amyloid plaques in Alzheimer's disease with sensitivity down to picomolar concentrations.
| Method | Spatial Resolution | Temporal Resolution | Advantage |
|---|---|---|---|
| fMRI | 1–3 mm | 1–2 s | Whole brain, noninvasive |
| PET | 2–4 mm | Minutes | Molecular markers (metabolism, receptors) |
| MEG | 2–5 mm | 1 ms | High temporal resolution, noninvasive |
| EEG | 5–10 cm | 1 ms | Portability, low cost |
Magnetoencephalography (MEG) records magnetic fields of 10–100 femtotesla generated by synchronous activity of 10⁴–10⁵ neurons, with temporal resolution of 1 ms and source localization accuracy of 2–5 mm.
Electroencephalography (EEG) measures summed electrical activity through 19–256 scalp electrodes, revealing rhythms: delta (0.5–4 Hz, deep sleep), theta (4–8 Hz, meditation), alpha (8–13 Hz, relaxed wakefulness), beta (13–30 Hz, active thinking), gamma (30–100 Hz, cognitive integration).
Patch-clamp technique achieves single ion channel resolution (conductance 1–100 picosiemens), allowing analysis of synaptic currents with amplitudes of 5–50 pA and microsecond precision — this is the boundary between macroscopy and molecular physics.
Multi-channel electrode arrays (up to 1024 channels) record activity from hundreds of neurons simultaneously in vivo, revealing population coding patterns and neural ensembles correlated with behavior.
Molecular and Genetic Approaches: Manipulating Neural Circuits
Optogenetics uses light-sensitive proteins (channelrhodopsin-2 activated by 470 nm blue light, halorhodopsin by 580 nm yellow light) to control activity of genetically defined neuronal populations with millisecond precision. Expression of opsins under specific gene promoters (e.g., CaMKIIα for excitatory neurons, GAD67 for inhibitory) allows selective activation or inhibition of cell types, proving their causal role in behavior.
Chemogenetics (DREADD — designer receptors exclusively activated by designer drugs) provides pharmacological control of neuronal activity: synthetic ligand clozapine-N-oxide (CNO, 1–10 mg/kg) activates inert receptors, modulating target neuron activity for hours. Viral vectors (AAV, lentivirus) deliver genetic constructs with 60–90% transduction efficiency to target areas of 0.5–2 mm³ volume.
- Optogenetics: light → opsin → ion channel → neuron active/inactive (milliseconds)
- Chemogenetics: CNO → synthetic receptor → activity modulation (hours)
- CRISPR/Cas9: genome editing → gene knockout → disease modeling (permanent)
- Viral vectors: construct delivery to target area (60–90% efficiency)
CRISPR/Cas9 genome editing enables creation of specific gene knockouts in neurons with 40–80% efficiency, modeling genetic variants associated with neurological diseases.
Single-cell transcriptomics (scRNA-seq) has revealed over 100 molecularly distinct neuronal types in cortex, each with unique expression profiles of 15,000–20,000 genes, redefining brain cell classification.
Connectomics reconstructs complete maps of synaptic connections: the C. elegans connectome (302 neurons, 7,000 synapses) was completed in 1986, a mouse cortical cube of 1 mm³ (100,000 neurons, 10⁹ synapses) in 2021 using serial electron microscopy and machine learning. This is the transition from description to complete topography.
Calcium imaging with GCaMP6 indicators (ΔF/F up to 1000% during activity) allows recording activity of 1,000–10,000 neurons simultaneously in vivo through implanted microscopes (1–2 g) or two-photon microscopy through cranial windows.
Cognitive Neuroscience: Neural Codes of the Mind
Cognitive neuroscience studies the neural mechanisms of higher mental functions—perception, attention, memory, language, decision-making, consciousness. The key concept is neural coding: information is represented by activity patterns of neuronal populations, where meaning is determined not by individual cells but by distributed activity of ensembles of 10²-10⁴ neurons.
Temporal dynamics are no less important than firing rate: information is encoded in the precise timing of spikes relative to hippocampal theta rhythm (4-8 Hz) with accuracy of 10-20 ms.
Mechanisms of Memory and Learning: From Synapses to Systems
Long-term potentiation (LTP)—a sustained enhancement of synaptic transmission by 100-400% following high-frequency stimulation (100 Hz, 1 s)—is considered the cellular mechanism of memory. LTP in the hippocampus requires activation of NMDA receptors, which pass Ca²⁺ only during simultaneous depolarization and glutamate binding (Hebbian coincidence detector).
Activation cascade: CaMKII → phosphorylation of AMPA receptors → increase in their number at the synapse (from 20 to 80 receptors within 15-30 min). Late-phase LTP (L-LTP, >3 h) requires de novo protein synthesis through activation of CREB-dependent transcription, increasing dendritic spine size by 30-60% and stabilizing changes for days to months.
| Process | Stimulation | Effect | Function |
|---|---|---|---|
| LTP | 100 Hz, 1 s | Enhancement by 100-400% | Learning, encoding |
| LTD | 1 Hz, 15 min | Weakening by 30-50% | Forgetting, refinement |
Long-term depression (LTD)—the opposite process of synaptic weakening during low-frequency stimulation—provides forgetting and refinement of neural circuits.
Memory consolidation transfers information from hippocampus to neocortex over weeks to months through reactivation of neural patterns during sleep. During slow-wave sleep, hippocampal "replay" reproduces daytime activity sequences in compressed time (6-20x compression), synchronizing with cortical spindles (12-15 Hz, 0.5-2 s) for information transfer.
Sleep disruption in the first 6 h after learning reduces consolidation by 30-50%, while optogenetic enhancement of reactivation improves memory by 20-40%.
Working memory maintains 4±1 items in prefrontal cortex through sustained neuronal activity (10-30 Hz) during seconds-to-minutes delays, modulated by dopamine (optimum at moderate levels, inverted U-curve). Episodic memory is encoded by hippocampal "place cells" (active in specific locations, field 20-50 cm) and "time cells" (encoding temporal intervals of 1-30 s), forming a cognitive map of space-time.
Neural Basis of Consciousness: Integration and Global Workspace
Global neuronal workspace theory (Dehaene, Changeux) posits that conscious perception arises when a distributed network of prefrontal, parietal, and temporal areas "ignites," broadcasting information through long-range cortico-cortical connections. Conscious stimuli evoke late activity (>300 ms) with global synchronization in the gamma range (30-80 Hz).
Subliminal stimuli generate only local early activity (<200 ms) without propagation. Damage to prefrontal cortex or thalamic nuclei (intralaminar nuclei) disrupts consciousness, confirming their role in global integration.
- Conscious stimulus → late global ignition (>300 ms)
- Subliminal stimulus → local early activity (<200 ms)
- Prefrontal cortex damage → integration disruption
- Thalamic nuclei damage → loss of consciousness
Integrated information theory (Tononi) quantitatively defines consciousness through Φ (phi)—a measure of causal integration of the system: high Φ requires balance between functional specialization of modules and their integration, maximized in the thalamo-cortical system.
Neural correlates of consciousness (NCC)—minimal neural mechanisms sufficient for specific conscious experience—are localized in posterior cortical "hot zones": extrastriate visual cortex (V2-V4) for visual awareness, temporo-parietal cortex for bodily self-awareness.
Binocular rivalry shows that V1 activity correlates with physical stimulus, while V4 and inferior temporal cortex correlate with subjective perception, indicating V4 as a critical zone for visual consciousness.
Anesthetics (propofol, sevoflurane) suppress consciousness by disrupting thalamo-cortical connectivity and reducing EEG complexity (perturbational complexity index PCI from 0.5 in wakefulness to 0.2 under anesthesia), without affecting local activity.
Vegetative state is characterized by preserved metabolic activity (50-60% of normal by PET), but fragmented functional connectivity, whereas minimally conscious state shows partial restoration of long-range connections and responses to commands in 20-40% of cases.
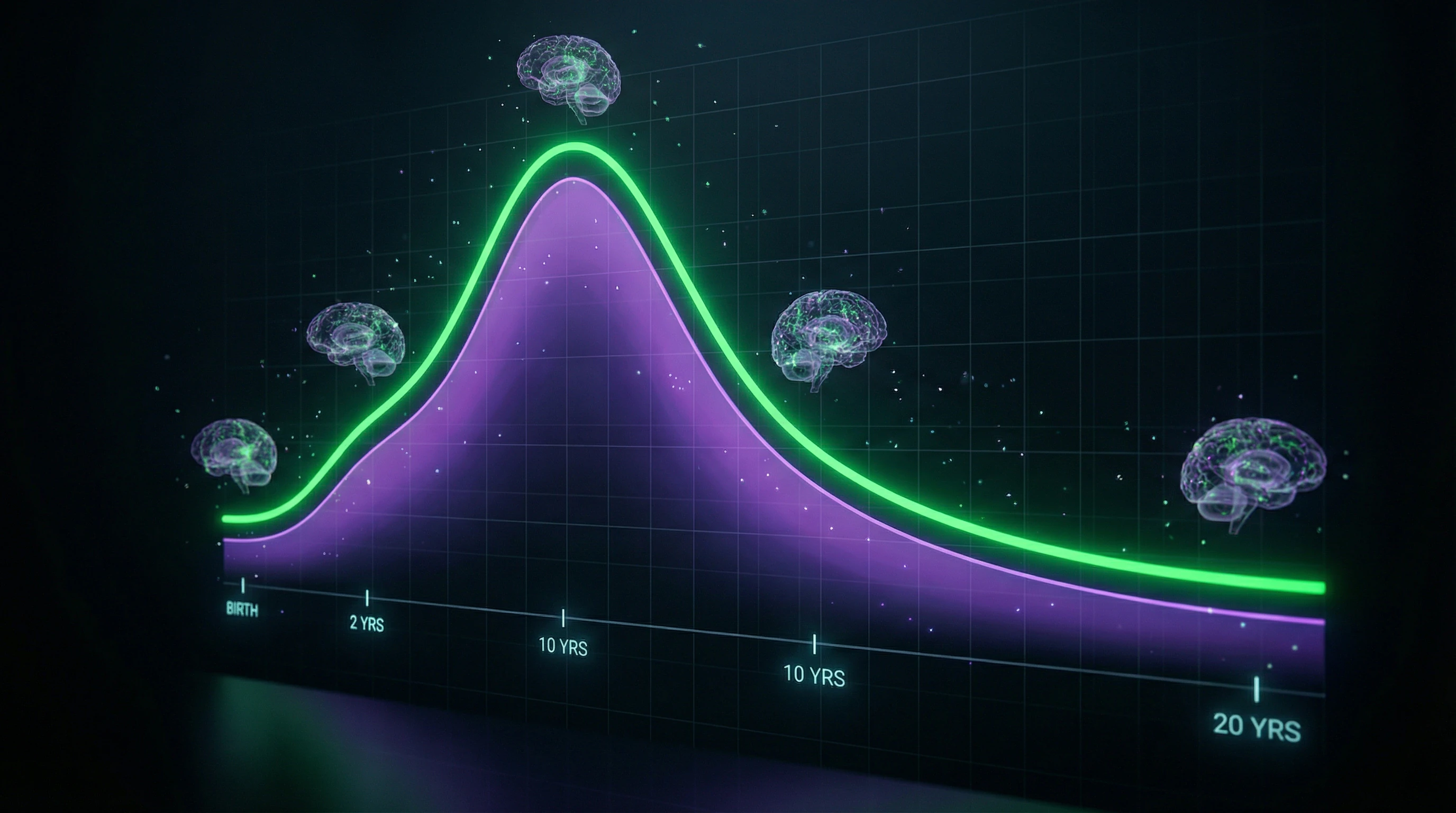
Neuroplasticity and Development — How the Brain Reorganizes Throughout Life
Formation of neural connections through synaptogenesis and pruning
Synaptogenesis peaks at different periods for different regions: visual cortex — 3–4 months (synapse density up to 150% of adult levels), prefrontal cortex — 1–2 years, followed by pruning that eliminates up to 40% of synapses by adolescence.
The mechanism depends on activity: frequently used synapses are stabilized through expression of BDNF and TrkB receptors, while inactive ones are tagged by complement C1q and eliminated by microglia.
- Long-term potentiation (LTP)
- Requires activation of NMDA receptors, Ca²⁺ influx, and phosphorylation of AMPA receptors, increasing synaptic strength by 200–400% within minutes with consolidation through protein synthesis over 1–3 hours.
- Enriched environment
- In rodents, increases dendritic spine density by 25%, hippocampal volume by 15%, and neurogenesis in the dentate gyrus by 50% compared to standard conditions.
Critical periods of brain development and their molecular mechanisms
The critical period for binocular vision in humans is 3–8 years, when monocular deprivation (strabismus, cataracts) causes irreversible amblyopia due to dominance of the open eye in visual cortex.
Opening and closing of critical periods is regulated by excitation/inhibition balance: maturation of parvalbumin-positive interneurons and perineuronal nets completes plasticity, while their degradation with chondroitinase ABC in adult rats restores juvenile plasticity.
Language development shows a critical period until age 7 for phonology (distinguishing native language sounds) and until age 15 for syntax, with progressive decline in accent-free second language acquisition ability of 0.7 proficiency points for each year of delay after age 7.
Adult neurogenesis persists in the subgranular zone of the hippocampus (700 new neurons/day in young adults, declining to 200/day by age 70) and subventricular zone, with integration of new neurons into existing networks over 4–6 weeks.
Clinical Neuroscience — Brain Pathologies and Their Neurobiological Foundations
Neurodegenerative diseases and protein aggregation mechanisms
Alzheimer's disease is built on two pathological pillars: extracellular β-amyloid plaques (Aβ42, oligomerizing at concentrations >100 nM) and intracellular neurofibrillary tangles of hyperphosphorylated tau protein (phosphorylation at 40+ sites instead of 2-3 normally). At the dementia stage, 30-40% of hippocampal and entorhinal cortex neurons are lost.
Pathology spreads according to the Braak staging scheme: stages I-II (entorhinal cortex, asymptomatic) → III-IV (hippocampus, mild cognitive impairment) → V-VI (neocortex, severe dementia). Correlation of tau pathology with cognitive decline (r=0.7-0.8) is significantly stronger than for amyloid (r=0.3-0.4).
| Disease | Primary mechanism | Key protein | Neuronal loss |
|---|---|---|---|
| Parkinson's disease | Death of dopaminergic neurons in substantia nigra | α-synuclein (Lewy bodies) | 60-80% by onset of motor symptoms |
| ALS | Selective degeneration of motor neurons | TDP-43 (97%) or SOD1 (20% familial) | 2-3% motor units per month; survival 3-5 years |
Parkinson's disease spreads according to the Braak hypothesis: from the olfactory bulb and dorsal motor nucleus of the vagus nerve to the midbrain and cortex. Amyotrophic lateral sclerosis (ALS) progresses at a rate of 2-3% motor unit loss per month, with median survival of 3-5 years from symptom onset.
Psychiatric disorders and their circuit-level neurobiology
Major depressive disorder is an imbalance of activity in the prefrontal-limbic network. The dorsolateral prefrontal cortex is hypoactive (15-25% reduction in metabolism on PET), the subgenual anterior cingulate cortex is hyperactive (Brodmann area 25, +30-40%), and the hippocampus shrinks by 8-10% in chronic depression due to glucocorticoid-induced atrophy.
The monoamine hypothesis explains the effect of SSRIs: they increase synaptic serotonin 3-5 fold, but therapeutic effects appear after 2-4 weeks because neuroplastic changes are required (increased BDNF, neurogenesis).
Schizophrenia demonstrates dopamine imbalance: the mesolimbic pathway is hyperactive (D2 receptors, +40-60% dopamine release → positive symptoms), the mesocortical pathway is hypoactive (prefrontal cortex → negative symptoms and cognitive deficits). Gray matter decreases by 2-3%, ventricles enlarge by 20-30%.
Obsessive-compulsive disorder (OCD) is characterized by hyperactivity of the orbitofrontal-striatal-thalamic-cortical loop: metabolism in the orbitofrontal cortex +25-35%, in the caudate nucleus +15-20%. After successful therapy (SSRIs or cognitive-behavioral therapy), activity normalizes.
Neurotechnology and the Future — Brain-Computer Interfaces and Neural Modulation
Brain-computer interfaces and neural activity decoding
Invasive BCIs with microelectrode arrays (Utah array, 96–128 electrodes) record action potentials from individual motor cortex neurons at 30 kHz resolution. This enables decoding of movement intention with 90–95% accuracy and control of robotic limbs with 7–10 degrees of freedom in real time (latency <100 ms).
Machine learning algorithms (recurrent neural networks, Kalman filters) transform activity patterns from 50–200 neurons into movement trajectories. Co-adaptive decoders learn through reinforcement, adjusting to signal changes.
- Non-invasive EEG-based BCIs use P300 (potential 300 ms after target stimulus, amplitude 5–10 μV) or motor imagery (desynchronization of 8–12 Hz μ-rhythm in contralateral motor cortex).
- Cursor or speller control achieves speeds of 5–25 characters/minute with 70–90% accuracy.
- Electrocorticography (ECoG) with subdural electrodes provides intermediate resolution (1–2 cm, 1 kHz), decoding speech with 75–90% accuracy for limited vocabulary and 40–60% for free speech.
Neuromodulation and therapeutic applications of brain stimulation
Deep brain stimulation (DBS) of the subthalamic nucleus in Parkinson's disease (130–185 Hz, 2–4 V, 60–90 μs) improves motor symptoms by 40–70% on the UPDRS scale and reduces levodopa requirements by 50–60%. Efficacy persists >10 years in 70% of patients.
Mechanism: suppression of pathological β-oscillation (13–30 Hz) in basal ganglia and normalization of thalamo-cortical activity. Adaptive DBS, which adjusts stimulation based on local field potentials (LFP), increases efficacy by 20–30%.
Transcranial magnetic stimulation (TMS) of the dorsolateral prefrontal cortex (10 Hz, 3000 pulses/session, 20–30 sessions) shows depression remission in 30–40% of patients resistant to antidepressants. The mechanism includes LTP-like plasticity and 20–35% increase in BDNF.
| Method | Parameters | Effect |
|---|---|---|
| tDCS (anodal) | 1–2 mA, 20–30 min | Increases cortical excitability by 30–40% |
| tDCS (cathodal) | 1–2 mA, 20–30 min | Decreases excitability by 20–30% |
| tDCS + working memory | Stimulation of corresponding areas | Improvement of 10–20% |
| tDCS + motor learning | Stimulation of corresponding areas | Acceleration of 15–25% |

Knowledge Access Protocol
FAQ
Frequently Asked Questions
Neuroscience is an interdisciplinary field that studies the structure, function, and development of the nervous system. It integrates biology, psychology, chemistry, and physics to understand how the brain works, the mechanisms of behavior, and cognitive processes. Research spans levels from molecules to entire neural networks.
Neurons transmit information through electrical impulses and chemical signals at synapses. When an impulse reaches the neuron's terminal, neurotransmitters are released and bind to receptors on the next cell. This process takes milliseconds and underlies all brain functions.
Neuroplasticity is the brain's ability to change its structure and function in response to experience and learning. Neural connections can strengthen, weaken, or form anew throughout life. This property allows the brain to recover from damage and adapt to new conditions.
No, this is a common myth debunked by neuroimaging. Research shows that all brain regions are active and perform specific functions, even at rest. Different tasks activate different areas, but throughout the day the entire brain is engaged.
MRI shows brain region activity but doesn't "read" specific thoughts. Functional MRI detects blood flow changes associated with neural activity, allowing identification of which areas are working during tasks. Decoding complex thoughts remains beyond current technology.
Chronic alcohol abuse can cause irreversible neuron damage, especially in the hippocampus and prefrontal cortex. However, with moderate consumption and cessation, the brain can partially recover through neuroplasticity. The degree of recovery depends on the duration and severity of use.
Regular physical exercise, quality sleep, and spaced repetition of information are effective. Aerobic activity stimulates neurogenesis in the hippocampus, while sleep consolidates memories. Mnemonic techniques, social engagement, and learning new skills that strengthen neural connections also help.
Neurotransmitters are chemical substances that transmit signals between neurons across synapses. Dopamine regulates motivation and pleasure, serotonin affects mood, and GABA inhibits neural activity. Neurotransmitter imbalances are linked to depression, anxiety, and other disorders.
Interfaces record the brain's electrical activity through electrodes and convert it into commands for devices. The technology uses EEG or implanted sensors to decode user intentions. Applications include controlling prosthetics, enabling communication for paralyzed patients, and stroke rehabilitation.
Yes, critical periods are time windows of maximum brain sensitivity to specific stimuli. For example, visual development is most active in the first years of life, and language skills are best acquired before adolescence. After these windows close, learning remains possible but less efficient.
This is an oversimplification that doesn't reflect reality. Both hemispheres participate in most cognitive processes, though they have some specialization. For example, the left is more often associated with speech, but creativity requires interaction between both hemispheres. Neuroimaging shows complex activity during any tasks.
Alzheimer's disease is characterized by accumulation of beta-amyloid plaques and tau protein in the brain, leading to neuronal death. Areas responsible for memory and thinking are affected, especially the hippocampus and cortex. The process begins decades before symptoms appear and is currently irreversible.
The effectiveness of most "brain trainers" is limited and doesn't transfer to everyday tasks. Research shows that improvements are specific to the trained exercises. More beneficial are diverse intellectual activities, language learning, musical instruments, and physical activity.
Chronic stress causes hippocampal atrophy, impairing memory, and enlarges the amygdala, intensifying anxiety. Elevated cortisol damages neurons and suppresses neurogenesis. However, these changes are partially reversible with stress reduction, meditation, and therapy.
Consciousness is integrated information processing by distributed neural networks, especially in the cortex and thalamus. Theories include global workspace and integrated information, but complete understanding is lacking. Research examines neural correlates of consciousness during perception, attention, and self-awareness.
This is a theoretical concept far from realization due to brain complexity and lack of understanding of consciousness's nature. It requires complete mapping of 86 billion neurons and trillions of synapses, as well as reproducing their dynamics. Philosophical questions about personal identity and subjective experience remain unresolved.
