 💧 Water Memory
💧 Water MemoryDebunking Common Misconceptions About Water Chemistryλ
From hydrogen water to the water cycle — a scientifically grounded analysis of myths that persist even after formal chemistry education
Overview
Misconceptions about water chemistry are persistent: 🧬 even after completing chemistry courses, students confuse covalent bonds with hydrogen bonds, attribute "memory" to H₂O, or believe in magical properties of "structured" water. Research documents predictable error patterns — from high schoolers to college graduates. Intuitive models ("water = simple liquid") block understanding of molecular dynamics and require targeted correction, not formula repetition.
🛡️
Laplace Protocol: All claims are based on peer-reviewed systematic reviews and meta-analyses, prioritizing sources with high citation indices and rigorous methodology to separate scientifically validated facts from common myths.
Reference Protocol
Scientific Foundation
Evidence-based framework for critical analysis
Protocol: Evaluation
Test Yourself
Quizzes on this topic coming soon
Sector L1
Articles
Research materials, essays, and deep dives into critical thinking mechanisms.
 💧 Water Memory
💧 Water Memory⚡
Deep Dive
Common Misconceptions About the Water Cycle in Education
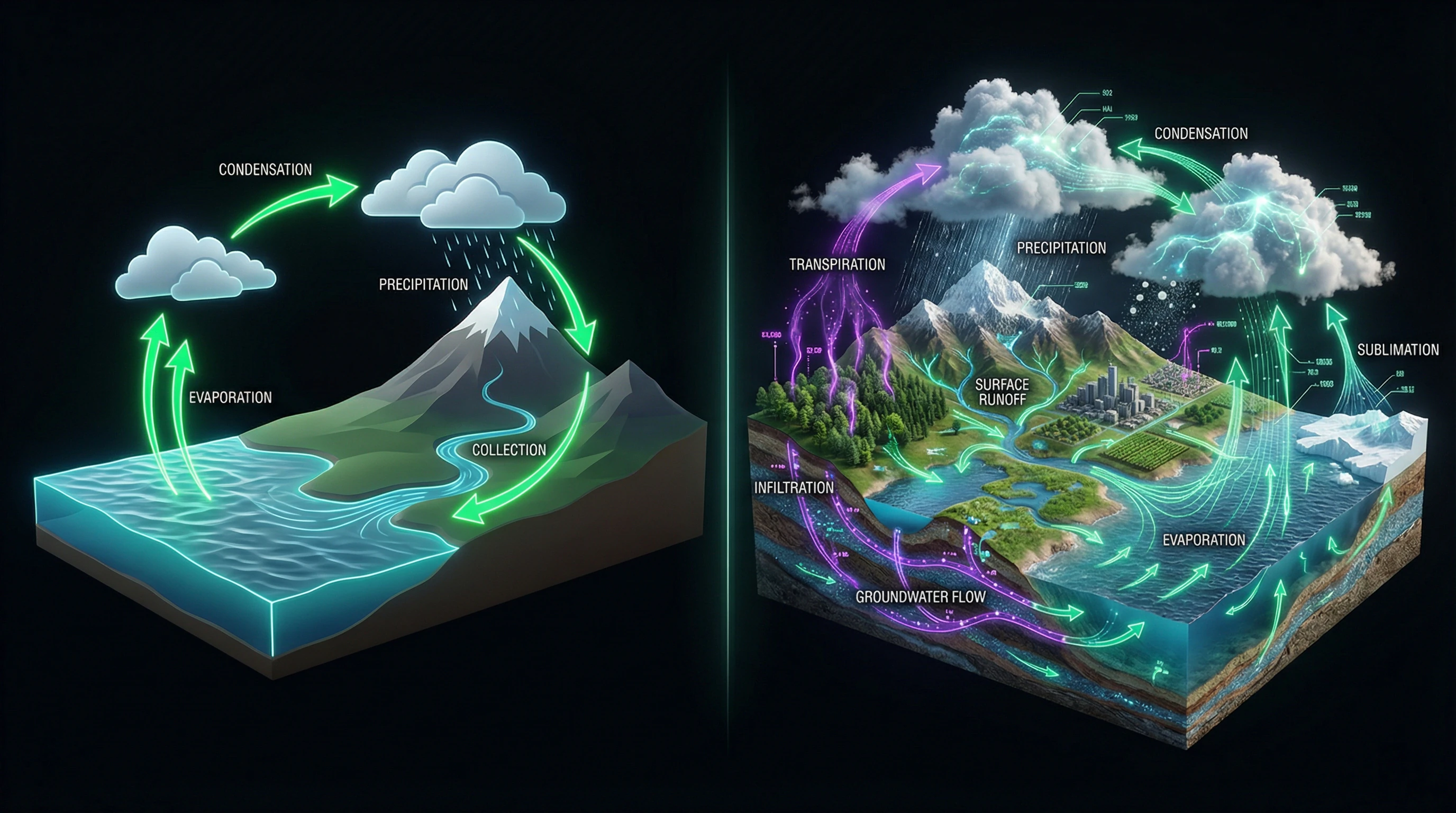
The water cycle is one of Earth's fundamental processes, but student understanding is often distorted by oversimplified models. Systematic research shows that misconceptions about the water cycle exist at all educational levels and persist even after formal instruction.
These errors are not random—they follow predictable patterns based on intuitive thinking and cognitive limitations in processing complex systems.
Oversimplification of Evaporation and Precipitation Processes
The most common misconception reduces the water cycle to a simple "water goes up and comes down" scheme. Students often represent the cycle exclusively as a sequence of evaporation and precipitation, ignoring intermediate stages.
Students rarely mention condensation as a separate process, merging it with cloud formation into a single undefined phenomenon. This oversimplification prevents understanding of energy transformations and water phase transitions.
Visual materials in textbooks often present cyclical diagrams with minimal elements, creating an illusion of simplicity. In reality, the process includes multiple parallel pathways and different time scales.
- Cognitive Load
- When processing the complete water cycle model, even students who know the correct information revert to simplified representations when solving problems or explaining phenomena.
Ignoring Groundwater and Transpiration
The underground component of the water cycle is systematically excluded from students' mental models. Groundwater, infiltration, and water movement through soil layers rarely appear in student descriptions, although these processes are critically important for understanding freshwater availability.
| Cycle Component | Status in Mental Model | Practical Significance |
|---|---|---|
| Surface Evaporation | Included in 95% of models | Visible process |
| Groundwater | Included in less than 30% of models | Primary freshwater source |
| Plant Transpiration | Included in less than 25% of models | Comparable to direct evaporation |
Transpiration—the release of water vapor by plants—remains an "invisible" process for most learners. Students do not realize that vegetation returns significant volumes of water to the atmosphere, comparable to evaporation from water bodies.
Failure to understand the role of forests in regional climate and water balance leads to underestimation of ecological risks from deforestation. Multiple mental models can coexist in a single learner—scientifically correct and simplified intuitive—activating in different contexts.
Myths About Hydrogen Water and Its Health Benefits
Hydrogen water — a product saturated with molecular hydrogen (H₂) — is actively promoted as a remedy with antioxidant and therapeutic properties. Marketing claims promise improved metabolism, slowed aging, and disease prevention.
Systematic reviews of clinical studies demonstrate limited and contradictory evidence for these assertions.
Systematic Review of Scientific Evidence
Meta-analysis of hydrogen water research reveals serious methodological problems. High-quality randomized controlled trials (RCTs) with adequate sample sizes are virtually nonexistent.
- Most studies were conducted on animal models or under in vitro conditions
- Results cannot be extrapolated to the human body
- Protocol pre-registration is absent
- Sample sizes are small (often fewer than 30 participants)
- Observation periods are short
Publication bias compounds the picture: studies with negative results are published less frequently, creating a distorted impression of effectiveness.
Quantity of studies does not compensate for their low quality — methodological rigor and reproducibility of results are critical for reliable conclusions.
Marketing Versus Reality
Commercial claims often rely on selective interpretation of preliminary data. Manufacturers cite studies on cell cultures or animals, presenting them as evidence of human benefits.
Bioavailability of molecular hydrogen through oral intake remains questionable — H₂ rapidly diffuses from the gastrointestinal tract, and its tissue concentration may be insufficient for claimed effects.
- Scientific Hypothesis
- Preliminary data requiring verification in controlled human trials
- Proven Therapeutic Effect
- Result confirmed by high-quality RCTs with adequate sample size and long-term observation
Consumers must distinguish between these categories. The hydrogen water phenomenon illustrates the gap between preliminary research and clinically significant evidence — a problem characteristic of science popularization in general.
Misconceptions About Water's Molecular Structure in Chemistry Education
The molecular structure of water and the nature of hydrogen bonds are fundamental chemistry topics, yet this is precisely where persistent errors concentrate. Research shows that misconceptions about water structure are present even among university students and can persist in practicing professionals.
These errors are not random—they reflect systemic problems in teaching molecular chemistry and chemical bonding.
Misunderstanding Hydrogen Bonds
Students often confuse hydrogen bonds with covalent bonds or represent them as weak electrostatic interactions without specific directionality. Many don't understand the partially covalent character of hydrogen bonds and their critical role in determining water's properties.
Common misconception: a hydrogen bond forms between hydrogen atoms of two molecules. In reality, it forms between the hydrogen of one molecule and the electronegative atom of another.
Visualization in educational materials often exacerbates the problem. Schematic diagrams create the impression of a static structure, whereas hydrogen bonds are dynamic—constantly breaking and reforming.
- Hydrogen Bond Dynamics in Liquid Water
- Each molecule forms an average of 3–4 hydrogen bonds, creating a three-dimensional network with constantly changing topology. The lifetime of a single bond is picoseconds.
- Why This Matters
- Failure to understand dynamics prevents explanation of water's anomalous properties: high heat capacity, surface tension, and density anomaly upon freezing.
Myths About Structured Water
The pseudoscientific concept of "structured" or "hexagonal" water with special biological properties has no scientific basis. Claims about long-lived ordered structures contradict thermodynamics—hydrogen bonds exist for picoseconds, and macroscopic ordering is impossible at room temperature.
Water does form temporary clusters, but their lifetime is measured in femtoseconds—this is a real phenomenon that pseudoscience reinterprets as "long-lived structures."
Commercial products promising "structuring" through magnetic fields or special treatment use scientific terminology to create an illusion of legitimacy. Critical analysis reveals an absence of reproducible experimental data confirming stable changes in water structure after such treatments.
- Student doesn't understand fundamental principles of molecular dynamics
- Becomes vulnerable to pseudoscientific concepts
- Multiple mental models (scientific and intuitive) coexist in memory
- Activated depending on context and cognitive load
Educational problem: a student may give the correct answer on an exam but apply the intuitive model when purchasing "structured water" in real life.
Myths About Water Pollution and Quality: The Invisible Threat
A common misconception: water contamination is always visible or detectable by taste. In reality, per- and polyfluoroalkyl substances (PFAS), heavy metals at low concentrations, and microbiological pathogens remain completely imperceptible to the senses.
The absence of visible signs of contamination does not correlate with chemical safety. This error is based on the intuitive assumption of a direct link between sensory perception and actual quality—an assumption that contradicts analytical data from modern environmental chemistry.
Organoleptic perception (taste, smell, color) is not an indicator of safety, but merely a crude filter for obvious contamination. Dangerous substances often remain invisible precisely because they are chemically inert to our receptors.
Invisible Contaminants: PFAS and Micropollutants
PFAS—a class of more than 4,700 synthetic compounds with exceptionally stable carbon-fluorine bonds. This ensures their persistence in the environment and bioaccumulation in the body.
These substances are detected in drinking water worldwide at concentrations from nanograms to micrograms per liter—completely undetectable without liquid chromatography with mass spectrometry. Long-term PFAS exposure is linked to endocrine disruption, immune dysfunction, and increased risk of cancer.
- Students underestimate risks from substances that do not manifest organoleptically
- Educational programs rarely include toxicology of micropollutants
- Targeted pedagogical interventions are required to develop adequate understanding of chemical risks
Misconceptions About Water Purification Methods
Myth: boiling eliminates all contaminants. In reality, boiling inactivates microbiological pathogens but does not remove heavy metals, nitrates, PFAS, and organic pollutants. Water evaporation can even concentrate non-volatile contaminants.
Household activated carbon filters are effective against chlorine and some organic compounds, but demonstrate limited effectiveness against inorganic ions. They require regular replacement, otherwise bacterial colonies begin growing in the filter material itself.
- Universal purification method
- Does not exist. Technology selection must be based on the specific chemical composition of source water, not on intuitive notions of "purification" as a single process.
- Molecular specificity
- Different contaminants require different approaches. This misconception persists precisely because it contradicts the intuition about universality.
Why Chemical Misconceptions Demonstrate Such Resistance to Correction
The persistence of chemical misconceptions is not an information deficit, but a complex cognitive phenomenon: erroneous concepts coexist with scientific knowledge in mental models. Even after chemistry courses, students and instructors activate intuitive, unscientific representations under cognitive load or in non-standard contexts.
Scientific concepts are often absorbed as isolated facts for exams, without integrating into deep intuitive models formed by everyday experience. What's required is not simply information, but creating cognitive conflict and explicit comparison of alternative models.
Cognitive Mechanisms Preserving Erroneous Representations
Chemical misconceptions are based on intuitive heuristics that work in the macroscopic world but are inapplicable at the molecular level. The notion that substances "disappear" when dissolved reflects visual perception but contradicts the principle of conservation of mass.
Confirmation bias leads learners to interpret new information through the lens of existing misconceptions, selectively attending to data consistent with erroneous models.
Meta-analysis shows: misconceptions are especially resistant when based on direct sensory experience, reinforced by social environment, possessing internal logical consistency, and not causing cognitive dissonance in everyday life.
Neurocognitive studies with functional MRI demonstrate: when activating scientific concepts in students with persistent misconceptions, increased activity is observed in brain regions associated with suppressing intuitive responses. This indicates ongoing cognitive struggle between competing models.
The Role of Intuitive Thinking in Forming Chemical Myths
Intuitive thinking (Kahneman's System 1) is a system of fast automatic judgments that evolved to process information with limited resources. It relies on heuristics ("representativeness," "availability") that systematically err in chemical contexts.
- The "structured water" myth exploits the intuition: orderliness = improved properties (true for macro-objects, but ignores thermodynamics at the molecular level)
- Students anthropomorphize molecules, attributing intentions to them ("molecules want to fill their shell"), reflecting teleological thinking
- Pedagogical strategies activating analytical thinking (System 2) through metacognitive reflection are more effective than traditional exposition
Explicit comparison of intuitive and scientific models demonstrates greater effectiveness in overcoming persistent misconceptions than simply providing correct information.
Systematic Review Methodology as a Tool for Myth Debunking
Systematic reviews are a methodologically rigorous approach to synthesizing scientific data, qualitatively distinct from narrative reviews through the absence of subjective source selection and explicit inclusion criteria. They include pre-registration of protocols, exhaustive searches across multiple databases, independent quality assessment by multiple reviewers, and standardized data extraction.
In the context of debunking chemical myths, systematic reviews enable quantitative assessment of the strength of evidence, identification of publication biases, and detection of methodological flaws in studies cited to support pseudoscientific claims. Meta-analysis provides statistical pooling of results from multiple studies, increasing statistical power and detecting effects invisible in individual studies.
Critical Distinctions from Conventional Literature Reviews
Narrative reviews are susceptible to selective choice of studies confirming the author's hypotheses and lack explicit quality assessment criteria. Systematic reviews require a priori specification of the research question in PICO format (Population, Intervention, Comparison, Outcome), ensuring focus and reproducibility.
- Search Strategy
- Documented with precision down to specific search terms and Boolean operators, allowing other researchers to reproduce the search and verify completeness of literature coverage.
- Quality Assessment
- Conducted using validated instruments (Cochrane Risk of Bias scale, Newcastle-Ottawa Scale), providing objective evidence grading.
- Critical Significance
- Particularly important when evaluating claims about "miraculous" water properties, where low-quality studies often create an illusion of scientific support.
Minimizing Bias in Chemical Data Analysis
Publication bias—studies with positive results are more likely to be published than work with null or negative results. This poses a serious threat to the validity of conclusions in chemical sciences.
Systematic reviews employ multiple strategies to detect and correct bias: searching for unpublished data in clinical trial registries, funnel plot analysis, and statistical methods such as trim-and-fill analysis.
Selective outcome reporting bias is detected through comparison of published results with pre-registered study protocols—this is a key mechanism distinguishing rigorous science from pseudoscience.
In the context of myths about hydrogen water or structured water, systematic reviews often reveal that claims are based on a small number of low-quality studies with high risk of bias, while more rigorous studies fail to confirm the claimed effects.
The GRADE system (Grading of Recommendations Assessment, Development and Evaluation) provides a structured approach to assessing the quality of the body of evidence, considering risk of bias, inconsistency of results, indirectness of evidence, imprecision of estimates, and publication bias. This allows grading confidence in conclusions from "very low" to "high."
Knowledge Access Protocol
FAQ
Frequently Asked Questions
These are persistent incorrect beliefs about water properties and processes that contradict scientific evidence. They occur at all educational levels and persist even after instruction, as they're based on intuitive understanding. Misconceptions can coexist with correct knowledge and emerge under cognitive load.
The most common misconception is oversimplifying the cycle to 'water evaporates and falls as rain.' In reality, students often ignore groundwater, plant transpiration, and infiltration. These simplified models persist across different educational levels.
Systematic reviews show limited evidence for claimed health effects. Most assertions are based on marketing rather than quality clinical research. The gap between advertising promises and scientific reality is significant.
This is a pseudoscientific term without support in chemistry. Water molecules constantly move and rearrange, not maintaining long-term 'structures' at room temperature. Claims about special properties of such water contradict fundamental laws of thermodynamics.
Many contaminants, including PFAS, have no taste, odor, or color, yet are toxic. A common misconception is that clear-looking water is safe. Only laboratory testing can detect such contaminants.
Hydrogen bonds between H₂O molecules determine unique properties: high boiling point, surface tension, and ice density. Students often misunderstand their nature, confusing them with covalent bonds within the molecule. This is a key misconception in chemistry education.
No, different methods remove different types of contaminants. Boiling kills microbes but doesn't remove heavy metals or PFAS, which require specialized filters. The misconception about universal purification methods is widespread.
They're based on intuitive thinking and everyday experience, making them resistant to change. Cognitive mechanisms allow incorrect and correct beliefs to coexist. Even professors demonstrate misconceptions in certain cognitive tasks.
A systematic review uses a rigorous protocol, registration, and explicit criteria for study selection. A regular review can be subjective and selective. The systematic approach minimizes bias and ensures reproducibility of results.
Visual assessment and taste are insufficient—test strips are needed for basic parameters or laboratory analysis for accuracy. Many dangerous substances cannot be detected without specialized equipment. Regular testing is especially important for well water.
This is a myth — misconceptions are found at all levels, including advanced students and instructors. They emerge especially under cognitive load or in non-standard situations. The presence of misconceptions is not directly related to overall academic performance.
No, systematic reviews minimize but do not completely eliminate all types of bias. Publication bias and limitations in data access remain problematic. Review quality depends on methodological transparency and critical evaluation of sources.
Quality and homogeneity of data matter more than the number of included studies. A large number of low-quality studies can distort conclusions. Methodological heterogeneity requires careful interpretation of pooled results.
No, this is a common myth — misconceptions coexist with scientific knowledge and can resurface. Targeted pedagogical interventions are required to overcome them. Simply presenting correct information is insufficient for lasting conceptual change.
Students often confuse intramolecular O-H covalent bonds with intermolecular hydrogen bonds. Misunderstanding the dynamic nature of hydrogen bonds and their influence on properties is also widespread. These misconceptions are typical across all levels of chemistry education.
Everyday experience forms simplified models that work in daily life but contradict science. Intuition suggests heavy objects sink, yet ice floats — this requires understanding molecular structure. Such intuitive models follow predictable patterns and resist change.
