What is colloidal silver and why is it called a "natural antibiotic"—unpacking the terms and historical roots of the myth
Colloidal silver is a suspension of microscopic metallic silver particles in liquid, usually distilled water. Particle size ranges from nanometers to micrometers, with concentrations from a few parts per million (ppm) to hundreds of ppm in "enhanced" formulas. More details in the Essential Oils section.
Sellers position it as a universal antimicrobial agent for internal use, topical application, and even inhalation. Claims range from "immune support" to "fighting viruses" and "detoxification."
Historical substitution: from medical tool to dietary supplement
Before antibiotics, silver compounds were indeed used in medicine—silver nitrate was applied to cauterize wounds, and colloidal solutions were tried for syphilis and gonorrhea in the early 20th century. This creates an illusion of legitimacy: if silver was in medicine, it must work.
However, with the invention of penicillin in 1928, silver lost clinical significance due to high toxicity and unpredictable efficacy. Modern medicine uses silver-containing coatings for catheters and burn dressings—but this is topical application of controlled doses, not internal consumption of undefined concentrations.
Key substitution: the historical fact of silver use in medicine is transferred to modern colloidal silver for internal consumption, even though these are completely different applications with different risks.
What's sold as "colloidal silver" today
Composition varies: some products contain predominantly silver ions (Ag⁺), others metallic silver nanoparticles, still others a mixture of both forms with added protein stabilizers. None of these products have undergone clinical trials for internal use required for drug registration.
- FDA (U.S. Food and Drug Administration)
- Since 1999, officially states: colloidal silver is not safe or effective for treating any disease. This is not an opinion but a regulatory position based on the absence of evidence.
Scope boundaries: what this article covers and what it doesn't
We analyze claims about the benefits of colloidal silver for internal use and the risks of argyria. We do not examine topical application of silver-containing medical devices (dressings, catheters), where silver is used under controlled conditions and does not enter systemic circulation in significant amounts.
Also excluded are industrial applications of silver nanoparticles in textiles or packaging—where doses and exposure pathways are fundamentally different. This distinction matters: the same substance can be safe in one context and dangerous in another.

Seven Arguments from Colloidal Silver Advocates — The Steel-Man Version Without Straw Men
To honestly examine a myth, we must first present it in its most convincing form. Here are the strongest arguments made by colloidal silver proponents, formulated without distortion. For more details, see the section on Detox Myths.
⚙️ Argument 1: Antibacterial Activity of Silver Is Proven In Vitro
Laboratory studies do indeed show that silver ions and nanoparticles suppress bacterial growth in petri dishes. Research on biodegradable chitosan-based particles with colloidal silver demonstrated antibacterial activity against test cultures (S005). The mechanism involves damage to bacterial cell membranes and disruption of enzyme function.
Proponents argue: if silver kills bacteria in a test tube, it should work in the body too. The logic seems irrefutable at first glance.
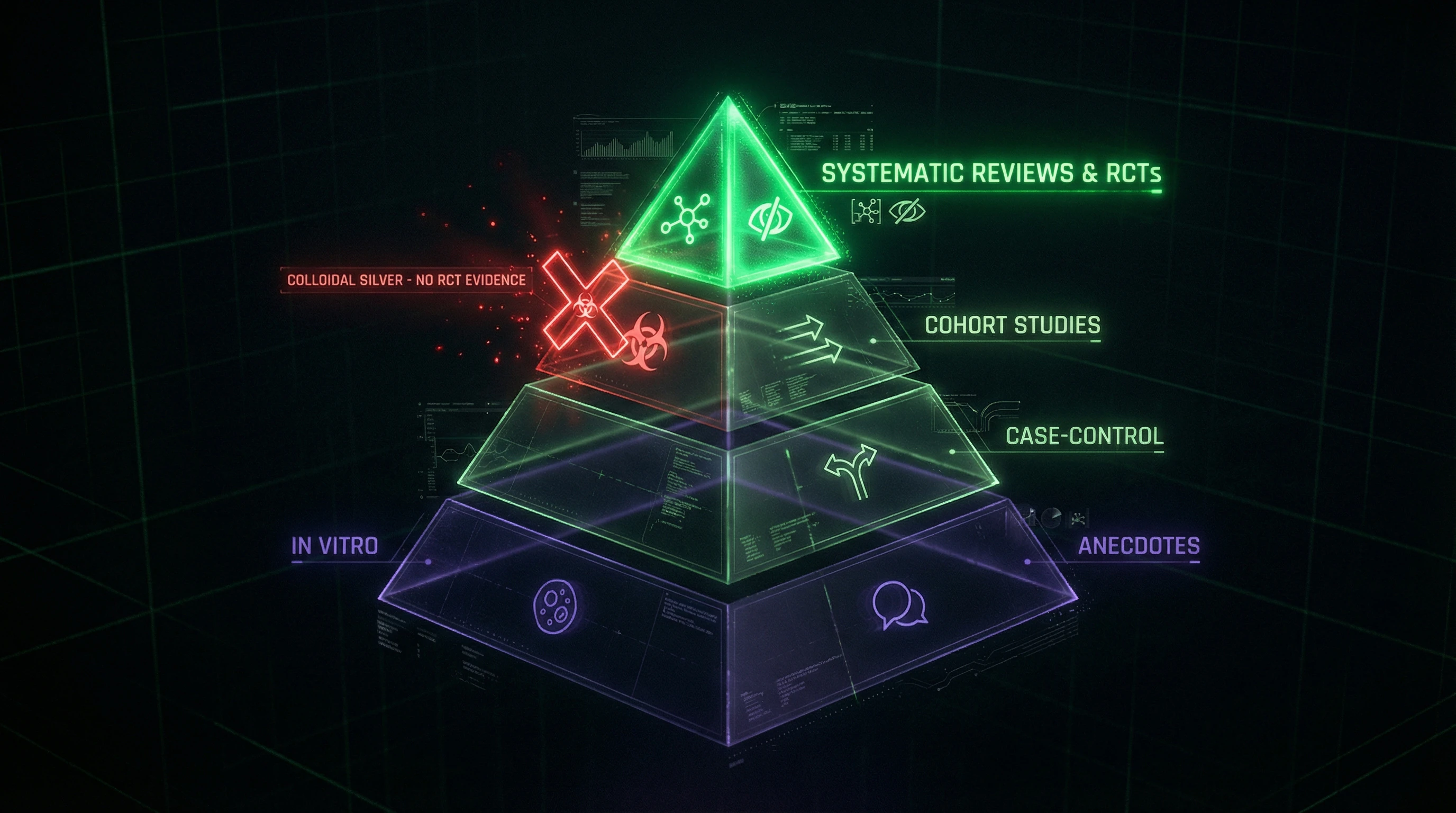
| Evidence Level | What's Shown | What It Means for Humans |
|---|---|---|
| In vitro (test tube) | Silver kills bacteria | Unknown — clinical trials needed |
| In vivo (animals) | Effect in animal organism | May not replicate in humans |
| Clinical trials | Effect in humans under controlled conditions | Applicable to humans |
⚙️ Argument 2: Silver Has Been Used in Medicine for Centuries
Historical evidence of silver use in medicine exists: ancient Greeks stored water in silver vessels, 19th-century physicians applied silver nitrate to treat wounds. Proponents interpret this as proof of safety and efficacy, appealing to "ancestral wisdom" and being "time-tested."
⚙️ Argument 3: Modern Medicine Uses Silver in Dressings and Catheters
Silver-containing burn dressings and silver-coated catheters are realities of clinical practice (S004). Proponents extrapolate: if medicine recognizes the antimicrobial properties of silver for external use, why not use it internally?
Here lies the trap: external application on a limited skin area is completely different from systemic introduction into the bloodstream.
⚙️ Argument 4: Personal Testimonials of Recovery
The internet is full of testimonials from people claiming colloidal silver cured their infections, colds, and skin conditions. These stories are emotionally convincing and create an illusion of massive evidence through survivorship bias — those who weren't helped or were harmed are less likely to write reviews, or their voices drown in the flood of positivity.
⚙️ Argument 5: Distrust of the Pharmaceutical Industry and Antibiotics
The problem of antibiotic resistance is real, antibiotic side effects exist, and pharmaceutical companies are indeed profit-motivated. Colloidal silver proponents position it as a "natural" alternative, free from corporate influence and the side effects of "chemicals."
This appeals to a real problem but draws a false conclusion: the existence of a problem doesn't make an ineffective solution effective. For more on cognitive traps in choosing "miracle cures," see the article on vitamin megadoses.
⚙️ Argument 6: Studies on Plants and Animals Show Efficacy
There are studies demonstrating the effectiveness of colloidal silver preparations in controlling plant diseases (S003). Proponents transfer these results to humans, ignoring differences in physiology and dosages.
- Extrapolation from Plants to Humans
- Plants lack the immune system of mammals, kidneys to excrete silver, or liver for detoxification. Result in plants ≠ result in humans.
- Extrapolation from Animals to Humans
- Even if silver works in rats, this doesn't guarantee safety and efficacy in humans. Clinical trials are required.
⚙️ Argument 7: Argyria — A Rare Exception from Abuse
Defenders of colloidal silver acknowledge the existence of argyria but present it as the result of extreme doses or improper use. They claim that with "reasonable" use, the risk is minimal and benefits outweigh risks.
The problem is that a "reasonable dose" is not defined by any regulatory body, and silver accumulation in the body is an individual and unpredictable process (S006).
What the Data Says: Evidence Review of Antibacterial Activity, Pharmacokinetics, and Clinical Efficacy
Let's examine each link in the chain of claims through the lens of scientific data, not limiting ourselves to laboratory experiments. More details in the Alternative Oncology section.
📊 In Vitro Activity vs. Clinical Efficacy: Why a Petri Dish Isn't a Human
Yes, silver kills bacteria under laboratory conditions (S001). But the path from test tube to clinic is littered with failures.
When colloidal silver enters the gastrointestinal tract, it encounters hydrochloric acid, digestive enzymes, and food proteins that bind silver ions and drastically reduce its bioavailability. Pharmacokinetic studies show that only a small fraction of ingested silver is absorbed into the bloodstream; most is excreted in feces. Blood silver concentrations after typical doses are orders of magnitude lower than concentrations that demonstrated antibacterial effects in vitro.
📊 Absence of Randomized Controlled Trials in Humans
As of 2026, there exists not a single quality randomized controlled trial (RCT) demonstrating the efficacy of oral colloidal silver for treating infections in humans. All claims are based either on in vitro data, animal/plant studies (S003), or anecdotal evidence.
This is a critical failure of the evidence base: without RCTs, it's impossible to separate placebo effect from real action, impossible to assess safety and dose-dependence.
- No control group — no comparison with placebo or standard treatment.
- No blinding — patient and physician know silver is being taken.
- No randomization — impossible to exclude selection bias.
- No long-term monitoring — delayed effects unknown.
🧾 Adsorption Properties and Interaction with Biological Molecules
Studies of adsorption properties in silver-cholesterol hybrid systems show that silver nanoparticles actively interact with lipids and proteins (S005). In the body, this means silver will bind to blood proteins, cell membranes, lipoproteins — but not selectively to bacteria.
Such nonspecific binding reduces antibacterial activity while simultaneously creating conditions for toxicity. Silver becomes not a weapon against infection, but ballast in the bloodstream.
🔬 The Standardization Problem: What's Actually in the Bottle
Analysis of commercial colloidal silver products reveals enormous variability: stated concentrations often don't match actual levels, particle size varies from batch to batch, the ratio of ionic to metallic silver is unpredictable.
| Parameter | Claimed Value | Actual Range | Consequence |
|---|---|---|---|
| Silver concentration | 10–50 ppm | ±40–60% | Impossible to assess dose |
| Particle size | 1–10 nm | Varies batch to batch | Different bioavailability and toxicity |
| Ionic vs. metallic silver | Not specified | Unpredictable | Different mechanisms of action and side effects |
Without standardization, it's impossible to speak of reproducible effects — what worked (or didn't work) in one case may be a completely different product in another bottle. This renders any conclusions about safety and efficacy unreliable.
The Mechanism of Argyria: How Silver Turns Skin Blue-Gray — Molecular and Cellular Anatomy of Toxicity
Argyria is not a cosmetic defect, but a marker of systemic silver accumulation in tissues. Understanding the mechanism is critical for risk assessment. Learn more in the Statistics and Probability Theory section.
🧠 Pharmacokinetics of Silver: Absorption, Distribution, Deposition
After oral ingestion, a portion of silver (predominantly in ionic form) is absorbed in the small intestine. In the bloodstream, silver ions bind to proteins, especially albumin and metallothioneins.
Silver distributes throughout organs, accumulating primarily in the skin, liver, kidneys, and spleen (S006). In the skin, silver deposits in the dermis, especially around sweat glands and hair follicles.
| Process Stage | What Happens | Key Outcome |
|---|---|---|
| Absorption | Silver ions pass through the small intestine wall | Entry into bloodstream |
| Transport | Binding with albumin and metallothioneins | Distribution to organs |
| Deposition | Accumulation in skin, liver, kidneys, spleen | Systemic deposition |
| Photoreduction | Under light exposure, ions → metallic nanoparticles | Blue-gray skin discoloration |
The photoreduction process is identical to what occurs in silver-based photography: light reduces ions to metal, which gives the skin its characteristic color.
🔁 Irreversibility of Argyria: Why the Blue-Gray Discoloration Doesn't Fade
Metallic silver deposited in the skin is chemically inert and is not eliminated by the body (S004). The half-life of silver elimination from the body is weeks to months, but particles already deposited remain permanently.
Argyria is a lifelong mark. Laser therapy can partially lighten the skin by breaking down large silver aggregates, but complete restoration of normal skin color does not occur.
🧬 Dose-Dependence and Individual Variability
No clear safe dose threshold exists. Cases of argyria have been documented after cumulative intake of 4 to 40 grams of silver over months to years (S006).
- Genetic differences in metal metabolism
- Liver and kidney function
- Concurrent intake of other substances
- Individual skin sensitivity
These factors affect accumulation rates and make it impossible to establish a "safe dose" for the general population.
🧷 Other Toxic Effects of Silver: Not Just Cosmetic
Beyond argyria, chronic silver exposure has been linked to neurological disorders (brain deposition), impaired kidney function, and liver changes (S002). Data is limited due to the rarity of prolonged high-dose intake, but cases are documented.
Silver is a heavy metal, and its accumulation in the body does not occur without consequences. This fundamentally distinguishes colloidal silver from substances the body can metabolize or eliminate.
Anatomy of Cognitive Traps: Which Mental Biases Make the Colloidal Silver Myth So Persistent
The psychological mechanisms sustaining belief in colloidal silver operate independently of biochemistry. They're embedded in how the brain processes information under uncertainty and social pressure. More details in the Mental Errors section.
🧩 Appeal to Nature: Natural = Safe
Silver is an element on the periodic table, a metal that exists in nature. Marketing exploits a cognitive bias: natural = good, ignoring that arsenic, mercury, and cyanide are also natural.
Nature isn't optimized for human health. Evolution hasn't equipped us with mechanisms to safely metabolize silver in gram quantities—just as with most heavy metals.
🧩 Survivorship Bias and Confirmation Bias
People for whom colloidal silver "worked" (or whose infection resolved on its own, which happens in most viral respiratory cases) actively share their experiences. Those who noticed no effect or were harmed stay silent.
This creates an illusion of high effectiveness. Confirmation bias causes proponents to notice cases of "success" and ignore failures—a mechanism identical to what operates with vitamin megadoses and other "miracle cures."
🕳️ Fear of Antibiotics and Corporations
Real problems—antibiotic resistance, drug side effects, pharmaceutical companies' commercial interests—create grounds for distrust. Colloidal silver is positioned as an "independent" alternative, which is emotionally appealing.
But replacing a regulated, studied product with an unregulated, unstudied one isn't a solution. It's jumping from the frying pan into the fire, where unknown risks (S006) outweigh hypothetical benefits.
🧩 Extrapolation Error: From Test Tube to Human
Proponents make a logical leap: silver works in vitro → therefore it will work in vivo; silver is used in wound dressings → therefore it's safe internally (S004).
- Each transition requires separate proof.
- Concentration, form, and route of administration radically change the effect.
- Absence of evidence isn't evidence of absence, but neither is it grounds for action.
This is a classic generalization error without accounting for context, which also appears in turmeric myths and other areas of pseudomedicine.
The persistence of the colloidal silver myth isn't in its truth, but in how it simultaneously appeals to fear of the system, desire for control, and cognitive laziness: it's easier to believe in one remedy than to understand the complexity of immunology and pharmacology.
The "Miracle Cure" Verification Protocol: Seven Questions That Expose 90% of Pseudoscientific Claims in Two Minutes
A universal checklist for evaluating any health claims, applicable far beyond the topic of colloidal silver. More details in the Feng Shui and Vastu section.
- Are there randomized controlled trials on humans? If the answer is "no" or "only in vitro / on animals" — red flag. RCTs are the gold standard of evidence (S002). Without them, efficacy claims are speculation.
- Are studies published in peer-reviewed journals with impact factors? Publication on a manufacturer's website, in an "alternative" journal without peer review, or in conference proceedings — not evidence. Check PubMed, Scopus, Web of Science databases.
- Who funded the study and are there conflicts of interest? Research paid for by the product manufacturer requires special caution (S006). Look for independent replications of results.
- Does it claim effectiveness against a broad spectrum of unrelated diseases? If a product "treats" infections, cancer, diabetes, and autoimmune diseases — that's a sign of quackery. Real medications have narrow, specific mechanisms of action.
- Does it use phrases like "natural," "chemical-free," "what doctors don't want you to know"? These markers are manipulation signals. Science doesn't divide substances into "chemical" and "natural" — everything consists of molecules. Conspiracy narratives replace evidence with emotion.
- Does it warn about possible side effects and contraindications? If a product is presented as absolutely safe for everyone — that's a lie. Any biologically active substance has side effects (S004). Honesty about risks is a sign of scientific approach.
- Is the product approved by regulators (FDA, EMA, national health authorities)? Lack of approval doesn't always mean ineffectiveness (new methods take time), but for products sold for decades without approval, it's a warning sign. The FDA has explicitly warned against colloidal silver since 1999.
If a product passes all seven questions without red flags — it's not a guarantee of safety, but reason to take it more seriously. If it fails on three or more — trust in it should drop below zero.
This protocol works not because it exposes lies, but because it reveals the absence of verification mechanisms. Pseudoscience doesn't fear questions — it fears answers to them.
Applying this checklist to colloidal silver, you get four red flags in a row: no RCTs on humans, funding comes from manufacturers, claims cover the entire spectrum of infections, uses "natural" and "hidden" rhetoric. This isn't coincidence — it's a pattern.
Similar analysis applies to megadoses of vitamins, turmeric as a panacea, and other "miracle cures." The cognitive trap mechanism remains unchanged.

Data Conflicts and Zones of Uncertainty: Where Sources Diverge and Why This Matters for Honest Analysis
Scientific integrity requires acknowledging limitations and contradictions. This is where science differs from dogma: it doesn't hide gaps in knowledge, but points to them.
🧾 Discrepancies in In Vitro Antibacterial Activity Assessment
Different studies report varying effectiveness of silver against bacteria depending on particle size, concentration, bacterial strain, and medium composition. Study (S001) shows activity under specific conditions, but extrapolation to other conditions or strains is not always reproducible.
This is normal for science, but creates problems for definitive conclusions. When a colloidal silver proponent sees one study with positive results, they ignore that the outcome depends on parameters that are completely different in a living organism.
Laboratory activity ≠ clinical efficacy. This isn't a contradiction, but different questions.
🧾 Absence of Long-Term Epidemiological Data
We don't know the exact prevalence of mild argyria or subclinical silver accumulation among colloidal silver users because systematic monitoring doesn't exist. Most data comes from case reports of severe argyria that made it into medical literature (S006).
Mild cases may go unnoticed or not be connected to silver intake. This creates systematic bias: we only see the tip of the iceberg.
🧾 Variability in Individual Sensitivity
Why do some people take colloidal silver for years without visible argyria, while others turn blue within months? Genetic differences in metabolism, kidney function, diet, microbiome—everything affects silver accumulation.
- Polymorphisms in genes encoding metal transporters (e.g., SLC31A1)
- Kidney function and silver excretion rate
- Gut microbiota composition affecting bioavailability
- Concurrent intake of other substances competing for absorption
Science lacks data to predict who's at risk. This doesn't mean there's no risk—it means we can't quantify it.
🧾 Contradictions in Silver Nanoparticle Safety Assessment
Studies (S002) and (S004) diverge in conclusions about silver nanoparticle cytotoxicity at low concentrations. Some authors find effects at 1–10 μg/mL, others only at 50+ μg/mL.
The reason: different cell lines, different culture conditions, different measurement methods. This isn't error—it's the reality of biological variability.
- Extrapolation Problem
- Cytotoxicity in cell culture doesn't equal toxicity in organisms. The body has barriers, metabolism, immunity. But this doesn't mean in vitro data is useless—it points to mechanism.
- Dose Problem
- Colloidal silver contains nanoparticles, but also silver ions. Which component is toxic? Both? It depends on pH and medium composition (S003). This complicates interpretation.
🧾 Zones of Honest Uncertainty
There are questions science hasn't yet answered, and that's normal. Acknowledging uncertainty is a sign of scientific maturity, not weakness.
| Question | What's Known | What's Unknown |
|---|---|---|
| Minimum cumulative dose for argyria | Case reports exist, but no prospective studies | Exact threshold for different genotypes and health states |
| Reversibility of mild argyria | Severe argyria is irreversible; mild may partially regress | Timeline and completeness of recovery |
| Antibacterial activity in vivo | In vitro activity proven; clinical efficacy not proven | Silver concentration at infection site and its bioavailability |
| Microbiome interaction | Silver kills bacteria; long-term microbiome effects unclear | Microbiome recovery after discontinuation |
These zones of uncertainty aren't reason to believe in a miracle cure. They're reason to demand evidence before use. The burden of proof lies with proponents, not skeptics.
Compare with other miracle cure myths: megadoses of vitamins or turmeric as panacea—everywhere the same pattern: laboratory data that doesn't translate to clinic, and knowledge gaps filled with belief.
