Candida and Leaky Gut: Scientific Facts and Mythsλ
We examine the connection between fungal microflora and intestinal barrier dysfunction based on scientific evidence and clinical research
Overview
The term "leaky gut" has migrated from alternative medicine into scientific discourse — it represents a measurable phenomenon: increased intestinal permeability. Candida albicans, a common resident of the microbiota, can disrupt epithelial tight junctions 🧬 and trigger systemic inflammation when imbalanced. Clinical data link this to autoimmune conditions, metabolic disorders, and chronic inflammatory bowel diseases.
🛡️
Laplace Protocol: We separate established scientific facts about intestinal barrier dysfunction from popular oversimplifications, relying on peer-reviewed research and clinical data. It's important to understand the difference between proven mechanisms and hypotheses requiring further investigation.
Reference Protocol
Scientific Foundation
Evidence-based framework for critical analysis
Protocol: Evaluation
Test Yourself
Quizzes on this topic coming soon
⚡
Deep Dive
What Actually Happens to the Intestinal Barrier in Increased Permeability
Scientific Definition and Mechanisms of Barrier Disruption
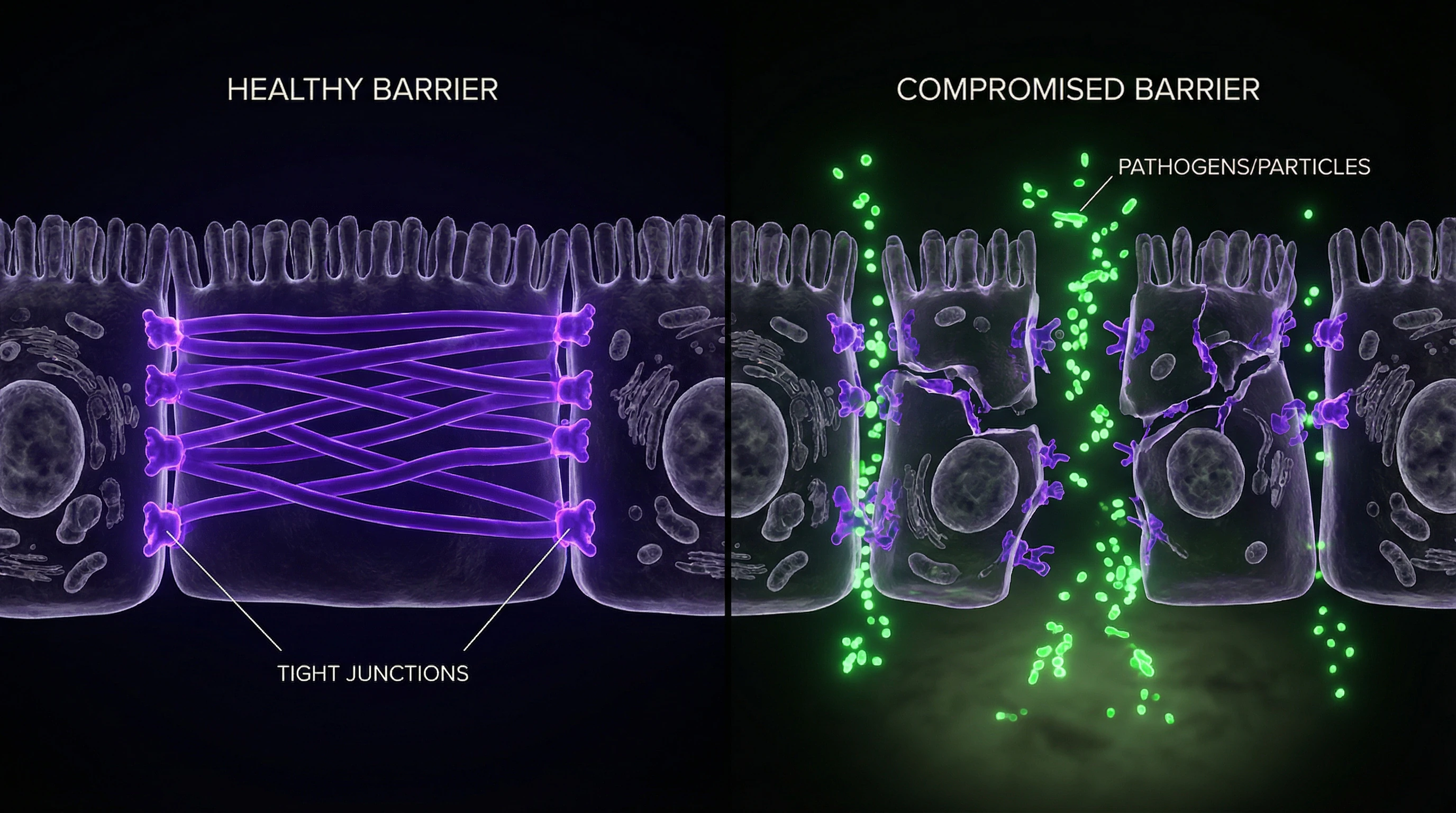
"Leaky gut" is a popular term for increased intestinal permeability, a condition in which tight junctions between epithelial cells weaken and allow substances from the intestinal lumen to penetrate into the bloodstream in increased amounts.
The intestinal barrier is a multi-layered system: a mucus layer, epithelial cells, tight intercellular junctions, and immune components of the mucosa. When disrupted, transport of lipopolysaccharides from gram-negative bacteria and other potentially toxic molecules across the intestinal wall increases.
| Barrier Component | Function | What Happens When Disrupted |
|---|---|---|
| Mucus layer | Physical protection, microbiota environment | Reduced thickness, loss of protection |
| Epithelial cells | Selective transport, metabolism | Apoptosis, dysfunction, increased permeability |
| Tight junctions | Regulation of paracellular transport | Disruption of connections, uncontrolled passage of molecules |
| Immune components | Pathogen recognition, tolerance | Hyperactivation or dysregulation |
Increased permeability is associated with chronic kidney disease, cardiovascular and dermatological problems. This is not an independent diagnosis, but a pathophysiological mechanism underlying multiple diseases.
Role of Tight Junctions Between Epithelial Cells
Tight junctions are a key element of barrier function. They form a selective barrier regulating paracellular transport (movement of substances between cells, not through them).
- Transmembrane proteins
- Occludin, claudins, JAM — span the membrane and create a physical seal between cells.
- Cytoplasmic proteins
- ZO-1, ZO-2, ZO-3 — link the complex to the cytoskeleton, providing mechanical strength.
- Structural disruption
- Any defect in these proteins leads to increased permeability and penetration of large molecules.
Pro-inflammatory cytokines, bacterial toxins, pH changes, and oxidative stress — all of these can disrupt the integrity of tight junctions.
When tight junctions become dysfunctional, bacterial components and undigested food antigens gain access to the submucosal layer, where they interact with immune cells. This initiates local and systemic inflammation, creating a vicious cycle: inflammation further impairs barrier function.
Candida in the Gut: From Normal Resident to Pathogen
Commensal Role of Fungi in the Microbiota
Candida albicans is a natural inhabitant of the intestine, part of the normal microbiota, and exists in balanced quantities without pathological consequences. The intestinal ecosystem relies on dynamic equilibrium between bacteria, fungi, viruses, and archaea.
Candida is kept in check by competitive interactions with bacterial flora and host immune surveillance. In small amounts, these fungi may participate in metabolic processes and immune system training.
- Intestinal pH environment
- Nutrient availability
- Antimicrobial peptide production by epithelium
- Immunoglobulin secretion
Conditions for Overgrowth and Transformation into Pathogen
Uncontrolled Candida proliferation begins with dysbiosis—disruption of microbial balance. Broad-spectrum antibiotics eliminate competing bacteria, freeing niches for opportunistic microorganisms.
During overgrowth, Candida transitions from yeast form to hyphal (mycelial) form—more invasive and capable of tissue damage.
Immune dysfunction is a critical factor in candidiasis development. A healthy immune system effectively controls fungal populations; its impairment in HIV infection, corticosteroid use, chemotherapy, or congenital immunodeficiencies creates conditions for invasion.
Additional triggers include: high simple carbohydrate diet (nutritional substrate for fungi), chronic stress, and hormonal changes. When favorable conditions arise, Candida initiates a cascade of pathological changes affecting not only the intestine.
Mechanisms of Candida's Impact on Intestinal Barrier Permeability
Direct Epithelial and Tight Junction Damage
Candida albicans attacks the intestinal barrier through multiple pathways simultaneously. The hyphal form physically penetrates between epithelial cells and invades them, mechanically disrupting monolayer integrity.
Fungi secrete proteolytic enzymes—secreted aspartyl proteases and phospholipases—that degrade tight junction proteins, especially occludin. Result: paracellular permeability increases.
- Candidalysin (cytolytic peptide toxin) forms pores in epithelial cell membranes, causing cell death
- Surface adhesins (Als3) enable attachment and induce endocytosis—fungi penetrate inside cells
- Biofilms create a protected environment, enhancing resistance to immune factors and antifungal agents
The combination of these mechanisms leads to structural barrier disruption and increased permeability to various molecules.
Role of Dysbiosis and Immune Response in Barrier Disruption
Candida overgrowth is simultaneously a consequence and cause of microbial imbalance. Dysbiosis means reduced microbiota diversity, decreased beneficial bacteria (especially short-chain fatty acid producers), and growth of opportunistic microorganisms.
Short-chain fatty acids, primarily butyrate, provide energy to colonocytes and regulate tight junction protein expression. During dysbiosis, their production drops—the barrier weakens.
The immune response to Candida overgrowth paradoxically exacerbates damage. Recognition of fungal patterns (Dectin-1, TLR2, TLR4) activates production of proinflammatory cytokines: TNF-α, IL-1β, IL-6, IL-17.
| Cytokine | Barrier Damage Mechanism |
|---|---|
| TNF-α, IFN-γ | Directly disrupt tight junction function, increasing permeability |
| IL-1β, IL-6, IL-17 | Sustain chronic inflammation, amplify immune activation |
Chronic inflammation creates a vicious cycle: increased permeability allows more antigens to penetrate the barrier, which amplifies immune activation and further damage.
Intestinal microbiota disruptions affect even distant outcomes—for example, spontaneous preterm births—demonstrating the systemic significance of the intestinal barrier.
Scientific Evidence and Research Limitations on Candida-Gut Permeability Connection
What Clinical Research Confirms
Increased intestinal permeability is a real clinical phenomenon, documented in chronic kidney disease, cardiovascular conditions, and dermatological disorders. This indicates the systemic significance of intestinal barrier function.
The role of microbiota in disease pathogenesis has received substantial scientific validation. The microbial-tissue complex influences inter-individual variability in health outcomes.
Candida albicans is recognized as a commensal organism capable of becoming pathogenic under certain conditions—particularly following antibiotic therapy and immune dysfunction.
Barrier damage mechanisms have been studied in detail: endogenous and exogenous factors disrupt tight junctions between epithelial cells, increasing transport of lipopolysaccharides from gram-negative bacteria into the bloodstream.
| Factor | Mechanism of Action | Evidence Status |
|---|---|---|
| TNF-α, IFN-γ | Direct disruption of tight junction proteins | Confirmed |
| Microbiota dysbiosis | Association with systemic diseases | Correlational data |
| Candida-induced inflammation | Vicious cycle of inflammation and permeability | Requires clarification |
Areas Requiring Further Investigation
Direct evidence of a specific connection between candidiasis and leaky gut syndrome remains limited. Most available data are correlational rather than causal in nature.
The term "leaky gut" is used in both scientific and pseudoscientific contexts, creating terminological confusion and complicating research interpretation.
Systematic reviews on this specific topic are scarce. Narrative reviews may be subject to author bias.
Specific pathways linking gut dysfunction to systemic diseases remain the subject of active investigation, particularly in the context of gut-organ axes.
The influence of maternal gut microbiota on distant outcomes such as preterm birth demonstrates the complexity of systemic interactions that have yet to be fully deciphered.
Randomized controlled trials are needed to evaluate the efficacy of therapeutic interventions aimed at restoring barrier function in candidiasis.

Risk Factors and Triggers for Intestinal Barrier Disruption in Candidiasis
Antibiotics and Immunosuppression
Broad-spectrum antibiotic therapy eliminates competing bacterial populations that normally keep fungal colonization in check. This is one of the most significant risk factors for Candida overgrowth in the gut.
Immunosuppressive conditions—HIV infection, chemotherapy, prolonged corticosteroid use, immunodeficiency syndromes—transform commensal Candida into its pathogenic form. Impairment of local mucosal immunity (reduced secretory IgA, Th17 response dysfunction) weakens defenses against fungal invasion.
The combination of antibiotic therapy and immunosuppression creates a synergistic effect, dramatically increasing the risk of systemic candidiasis and barrier damage.
Diet and Lifestyle
A diet high in refined carbohydrates and sugars provides the primary energy source for fungi. Chronic stress activates the hypothalamic-pituitary-adrenal axis, elevates cortisol, and alters microbiota composition.
Alcohol abuse directly damages the intestinal epithelium, disrupts tight junctions, and promotes dysbiosis. Insufficient dietary fiber intake reduces production of short-chain fatty acids, which are essential for epithelial barrier integrity and immune response regulation.
| Risk Factor | Mechanism of Action | Consequence for Barrier |
|---|---|---|
| Broad-spectrum antibiotics | Elimination of competing bacteria | Uncontrolled Candida growth |
| Immunosuppression | Reduction of local IgA and Th17 | Fungal invasion into epithelium |
| High-carbohydrate diet | Energy substrate for fungi | Candida proliferation |
| Chronic stress | Hypercortisolemia, dysbiosis | Increased permeability |
| Alcohol | Direct epithelial damage | Tight junction disruption |
| Dietary fiber deficiency | Reduced short-chain fatty acids | Weakened barrier and immunity |
Diagnosis and Evidence-Based Approaches to Intestinal Barrier Restoration
Clinical Assessment Methods
Diagnosing increased intestinal permeability remains challenging due to the absence of standardized tests and clear diagnostic criteria. Three main approaches are used in practice, but each has limitations.
| Method | Principle | Clinical Status |
|---|---|---|
| Lactulose-mannitol | Urinary sugar excretion ratio | Research use; limited availability |
| Serum zonulin | Permeability biomarker | Controversial specificity and significance |
| Stool microbiology | Quantitative Candida assessment | Standard for candidiasis diagnosis |
Key point: simple presence of Candida in stool is normal for healthy individuals. Diagnosis requires quantitative assessment of fungal load, not merely detection.
Evidence-Based Barrier Restoration Strategies
Antifungal therapy with fluconazole or nystatin is indicated only for documented Candida overgrowth and under medical supervision. Prescribing without microbiological confirmation is an error in diagnostic reasoning.
Probiotic strains Lactobacillus rhamnosus GG and Saccharomyces boulardii compete with Candida and support barrier function, but efficacy depends on strain and dose. This is not a universal solution.
Dietary interventions—limiting refined carbohydrates and increasing dietary fiber—modulate the microbiota and improve intestinal metabolic health. This is the foundational level that should be the first step.
Glutamine, butyrate, and nutrients supporting enterocyte energy metabolism show potential in restoring epithelial integrity, but require further clinical research to determine optimal protocols.
Comprehensive Protocol and Self-Treatment Risks
Intestinal barrier restoration requires elimination of triggering factors: unjustified antibiotic use, immune status correction, lifestyle modification. This is a systemic approach, not a targeted intervention.
Self-diagnosis and self-treatment without medical supervision carry real risks. Symptoms attributed to "leaky gut" and candidiasis may mask serious diseases requiring specific therapy.
Individualization of therapeutic protocols based on clinical presentation, laboratory data, and risk factors is key to effective barrier function restoration. Universal protocols do not exist.
Knowledge Access Protocol
FAQ
Frequently Asked Questions
This is a popular term for increased intestinal permeability, when tight junctions between intestinal cells become compromised. As a result, substances that normally shouldn't enter the bloodstream pass through the intestinal wall — bacterial toxins, undigested food particles. The scientific term is disruption of the structural-functional integrity of the intestinal barrier.
Candida overgrowth can contribute to intestinal barrier disruption, but it's not the sole cause. During dysbiosis, fungi release toxins and enzymes that damage the intestinal epithelium. However, direct causation requires additional research — this is a multifactorial process.
There are no specific symptoms — manifestations are nonspecific and include bloating, bowel irregularities, food intolerances, chronic fatigue. Skin problems, allergic reactions, and inflammatory processes in various organs may occur. Diagnosis is made only by a physician based on comprehensive examination, not isolated symptoms.
Yes, antibiotics destroy not only pathogenic but also beneficial bacteria, creating conditions for fungal growth. Candida is resistant to antibacterial drugs and occupies the freed ecological niche. Risk is especially high with prolonged or repeated courses of antibiotic therapy without probiotic support.
Diet is an important but not sole component of intestinal barrier restoration. Eliminating trigger foods (sugar, alcohol, refined carbohydrates) and adding anti-inflammatory nutrients helps, but a comprehensive approach is required. Addressing root causes is necessary: stress, infections, medications, under specialist supervision.
Clinical practice uses tests for zonulin (permeability marker), lipopolysaccharides (LPS), antibodies to intestinal wall components. The lactulose-mannitol test and calprotectin assessment are also used. However, standardized diagnostic protocols don't yet exist — result interpretation requires physician expertise.
No, Candida is present in the intestines of most healthy people as part of normal microbiota. Problems arise only with overgrowth against a background of weakened immunity, dysbiosis, or other factors. Fungal carriage doesn't equal disease — microbial balance is what matters.
Probiotics can support microbiota restoration and compete with Candida for resources, but aren't standalone treatment. Effectiveness depends on strains, dosages, and individual characteristics. Best results are achieved in combination with diet, trigger elimination, and when necessary, antifungal therapy.
Yes, chronic stress directly increases intestinal permeability through the brain-gut axis. Stress hormones (cortisol) disrupt tight junctions between epithelial cells and alter microbiota composition. This creates a vicious cycle: stress worsens the barrier, and a compromised barrier intensifies inflammation and stress response.
Yes, Candida uses simple carbohydrates (sugar, white flour) as its primary energy source for growth. A high-carbohydrate diet creates a favorable environment for fungal proliferation. However, complete carbohydrate elimination isn't required — balance and carbohydrate quality matter (preference for complex carbs with fiber).
Research shows a connection between increased intestinal permeability and certain autoimmune conditions, but causation has not been definitively proven. A compromised barrier may allow antigens through, triggering an immune response. However, it's unclear what comes first — permeability or the autoimmune process; this remains an area of active research.
No, self-diagnosis is unreliable — candidiasis symptoms are nonspecific and overlap with numerous other conditions. Popular "home tests" (such as the glass of water test) lack scientific validation. Diagnosis requires laboratory analysis (stool culture, PCR) and clinical evaluation by a gastroenterologist or infectious disease specialist.
Beneficial foods include those rich in glutamine (bone broth, meat), omega-3s (fatty fish), polyphenols (berries, greens), and zinc (pumpkin seeds). Fermented foods (sauerkraut, kefir) support the microbiota. Fiber from vegetables is important for feeding beneficial bacteria that produce short-chain fatty acids, which strengthen the barrier.
Timeframes are individual and depend on the degree of damage, underlying causes, and adherence to recommendations — ranging from several weeks to several months. The intestinal epithelium renews every 3-5 days, but restoring barrier function and microbiota requires more time. Consistency is key: eliminating triggers, following an anti-inflammatory diet, and supporting the microbiota.
Yes, systemic antifungal agents can disrupt microbiota balance, though to a lesser extent than antibiotics. They affect not only pathogenic fungi but also commensal microorganisms. Therefore, they are prescribed strictly when indicated, in courses, with concurrent probiotic support and under medical supervision.
Research suggests a possible connection through the gut-brain axis: dysbiosis and inflammation may influence neurotransmitters and mood. Candida toxins could theoretically affect the CNS when permeability is increased. However, direct evidence is limited — this is a promising research direction requiring careful interpretation.
