What Are Ayurvedic Products and Why They Became the Center of a Toxicological Scandal
Ayurveda — one of the oldest systems of Indian medicine, has gained popularity in the West in recent years alongside other complementary and alternative medicine (CAM) methods. Unlike allopathic medicine, the production and access to CAM products are extremely weakly regulated (S010).
This regulatory gap has created ideal conditions for the mass distribution of products marketed as "natural" and "safe," but containing dangerous concentrations of toxic metals. More details in the section Candida and Leaky Gut.
🔎 Scale of the Problem: From Local Poisonings to Global Crisis
Toxicity of ayurvedic products due to metal content is becoming an increasingly recognized public health problem (S010). Herbal formulas and complementary medicine are used by the majority of the world's population — meaning this problem affects not a marginal group of enthusiasts, but billions of people.
When regulation is absent and demand is high, toxicity becomes not a side effect, but a built-in feature of the market.
⚠️ Why "Natural" Doesn't Mean "Safe"
The central cognitive error is the "appeal to nature fallacy." Consumers automatically equate "herbal" and "ancient" with "safe," ignoring that many natural substances are powerful toxins.
Lead, mercury, and arsenic are also "natural" elements found in nature. But their presence in the body causes irreversible damage to the nervous system, kidneys, liver, and other organs (S001).
🧾 Specific Examples: Which Products Contain Dangerous Metals
Laboratory testing by government health agencies confirmed the presence of lead, mercury, and arsenic in several products (S011):
| Product | Detected Metals | Status |
|---|---|---|
| Haidyanath Sundary Sakhi tablets | Lead, arsenic, mercury | Laboratory confirmed |
| Sharmayu Som Pushpa | Lead, arsenic | Laboratory confirmed |
| Unjiha Pushpadhanva Rasa | Lead, arsenic, mercury | Laboratory confirmed |
| Baidyanath Rajahprawartini Bati | Lead, arsenic, mercury | Laboratory confirmed |
| Acineutra tablets | Lead, mercury | Laboratory confirmed |
| Flexy tablets | Lead, mercury | Laboratory confirmed |
Two additional products are linked to reported adverse effects but were not subjected to laboratory testing: SG Phyto Pharma Palsinurion (contains naga bhasma — lead and/or shuddha parada — mercury) and Kamini tablets (often labeled as containing shuddha parada or shuddha hingula — purified cinnabar/mercury) (S011).
- Naga bhasma
- An ayurvedic lead-based preparation traditionally used to treat skin diseases and digestive disorders. The problem: lead accumulates in the body and causes neurological damage even at low doses.
- Shuddha parada
- "Purified" mercury in ayurvedic tradition. The "purification" process does not remove toxicity; mercury remains a neurotoxin regardless of form.

Steel Man: Five Strongest Arguments from Defenders of Ayurvedic Metals
Before examining evidence of toxicity, it's necessary to honestly present the most compelling arguments from proponents of metal use in Ayurveda. This is not a straw man, but a steel man — the strongest possible version of the opposing position. For more details, see the section Folk Medicine vs. Evidence-Based Medicine.
🕰️ Argument from Tradition: Millennia of Practice as Proof of Safety
Defenders of Ayurveda argue that the medicinal use of metals has been practiced for thousands of years, and if these substances were truly dangerous, the tradition would not have survived to the present day. According to this logic, the long-term survival of the practice serves as empirical proof of its efficacy and safety.
Ayurvedic texts describe complex "purification" procedures (shodhana) of metals, which supposedly transform toxic substances into therapeutic agents.
⚗️ Argument from Alchemical Transformation: "Bhasmas" as Non-Toxic Forms of Metals
Proponents claim that metals in Ayurvedic preparations are not present in elemental form, but as "bhasmas" — products of repeated calcination and processing that alter the chemical structure of the metal. According to this theory, lead in the form of naga bhasma or mercury in the form of shuddha parada lose their toxicity while retaining therapeutic properties.
The process of preparing bhasmas includes up to 100 cycles of heating and cooling with various herbal extracts — supposedly this converts toxins into a "safe" state.
🌍 Argument from Cultural Relativism: Western Toxicology Doesn't Apply to Eastern Medicine
Some defenders of Ayurveda argue that Western safety standards and toxicological norms were developed for synthetic pharmaceutical drugs and cannot be applied to traditional plant-mineral formulas. They point out that Ayurvedic preparations operate within a different paradigm — balancing doshas (vata, pitta, kapha) rather than suppressing symptoms as in Western medicine.
Therefore, the evaluation criteria should be different.
💊 Argument from Dosage: "The Dose Makes the Poison"
Defenders cite Paracelsus's fundamental principle of toxicology: "All things are poison, and nothing is without poison; the dosage alone makes it so a thing is not a poison." They argue that the quantities of metals in Ayurvedic preparations are so small that they cannot cause toxic effects, especially with short-term use.
Moreover, they point out that many modern pharmaceutical drugs also contain potentially toxic substances in controlled doses.
📚 Argument from Selective Reporting: Western Media Exaggerates Rare Cases
Proponents of Ayurveda argue that cases of heavy metal poisoning represent rare exceptions associated with low-quality or counterfeit preparations produced by unscrupulous manufacturers. They point out that millions of people worldwide use Ayurvedic preparations without any adverse effects, but these cases don't make the news.
- Western regulators are supposedly interested in discrediting competing medical systems
- Media exaggerates rare cases to attract attention
- The pharmaceutical industry protects its interests through criticism of alternatives
- Absence of harm reports is interpreted as proof of safety
- Information selection creates an illusion of consensus about harmlessness
Evidence Base: What Laboratory Analyses and Epidemiological Studies Show
Systematic analysis of ayurvedic product samples from lead poisoning investigations presents an unambiguous picture: a toxicological catastrophe, not an exception. More details in the Detox Myths section.
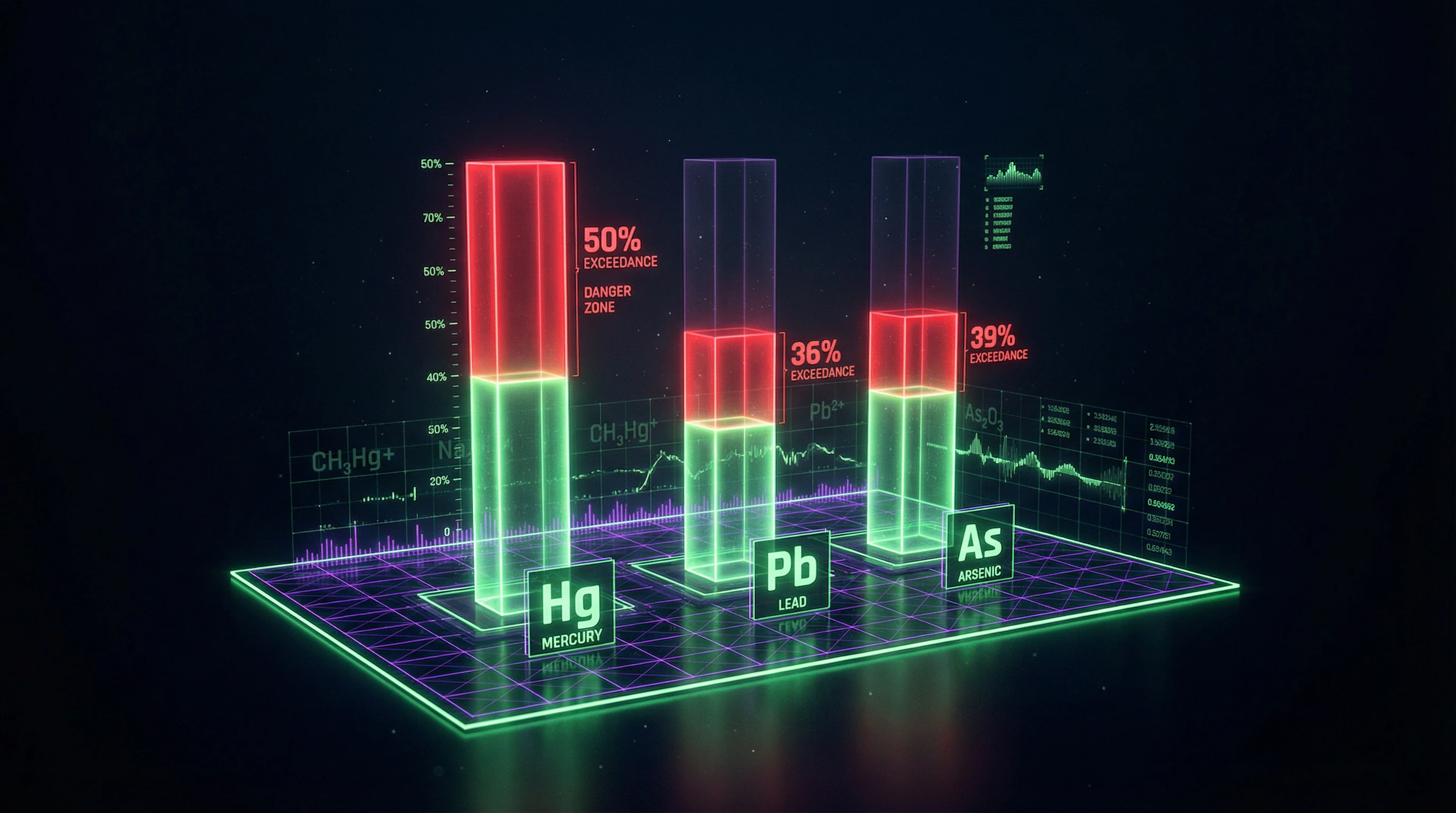
📊 Quantitative Data: How Many Products Exceed Safety Standards
Samples of ayurvedic formulas were analyzed for metal content according to U.S. standards (S010). Nearly half of samples containing mercury, 36% with lead, and 39% with arsenic had concentrations exceeding recommended daily limits for pharmaceutical impurities—in some cases by thousands of times (S010).
This is not measurement error or isolated incidents. This is a systematic problem affecting a significant portion of products on the market.
🧪 Measurement Methodology: How Dangerous Concentrations Were Determined
Reference limits for oral consumption are established by U.S. and European regulatory agencies (S010). Calculations used the United States Pharmacopeia (USP) standard, which provides guidelines for daily exposure to pharmaceutical impurities.
Conversion was performed from micrograms to milligrams (1 μg = 0.001 mg) based on a standard tablet weight of 500 mg and minimum dosage of one tablet per day for a 50 kg person (S010).
⚖️ Comparison with Safety Standards: How Large the Exceedances Are
- Lead: Permitted Daily Exposure (PDE) per USP—0.005 mg/day. Found above limits in more than one-third of samples (S010).
- Arsenic: PDE per USP—0.015 mg/day. Exceedances documented in 39% of samples (S010).
- Mercury: Nearly half of samples contained concentrations above safe levels.
Every third product represents a direct threat to health with regular use—not potential, but actual.
🔍 Concentration Variability: From Safe to Lethal
Half of samples had lead levels of 4.9 mg/kg and below (S010). Enormous variability between manufacturers and batches means that some products genuinely contain relatively low concentrations, while others are concentrated sources of toxins.
Consumers cannot distinguish safe products from dangerous ones without laboratory analysis. This creates the illusion of a lottery: each purchase is an unknown risk.
- Why Variability Is Not a Mitigating Factor
- The existence of "safe" batches does not solve the problem. Manufacturers do not control raw material sources and do not guarantee consistency. Buyers get roulette, not medicine. This violates the basic principle of pharmaceutical safety: dose predictability.
Mechanisms of Toxicity: How Lead, Mercury, and Arsenic Destroy the Body at the Molecular Level
Understanding the mechanisms of toxic action of heavy metals is critically important for assessing the real danger of Ayurvedic products. This is not an abstract threat, but specific biochemical processes leading to irreversible damage. More details in the Reality Check section.
🧠 Lead Neurotoxicity: Why Children and Pregnant Women Face Special Danger
Lead is a powerful neurotoxin, especially dangerous for the developing brain. It competes with calcium for binding to neurotransmission proteins, disrupts hemoglobin synthesis, and damages the blood-brain barrier.
In children, even low blood lead levels (below 5 μg/dL) are associated with reduced IQ, attention problems, and behavioral disorders (S001). Pregnant women and children face significantly higher risk: lead easily crosses the placenta and accumulates in fetal bones, creating a long-term reservoir of toxin.
Even after exposure ceases, lead continues to be released from bone tissue, maintaining chronic intoxication for decades.
💀 Mercury and Kidney Damage: From Acute Intoxication to Chronic Failure
Inorganic mercury (present in Ayurvedic products) primarily affects the kidneys. It accumulates in proximal tubules, causing necrosis of epithelial cells and acute kidney failure.
With chronic exposure, interstitial nephritis develops along with progressive decline in kidney function (S001). Mercury also has immunotoxic properties, triggering autoimmune reactions. Organic forms of mercury (methylmercury) are even more dangerous: they easily penetrate the blood-brain barrier and cause irreversible damage to the central nervous system.
- Acute Mercury Intoxication
- Manifests within hours to days: tremor, gingivitis, mental disorders, acute kidney failure.
- Chronic Mercury Intoxication
- Develops over months to years: microtremor, emotional lability, nephrotic syndrome, progressive kidney failure.
🔥 Arsenic and Carcinogenesis: The Mechanism of Cancer Induction
Arsenic is a proven human carcinogen (Group 1 by IARC classification). It induces oxidative stress, damages DNA, disrupts DNA repair, and alters epigenetic gene regulation (S001).
Chronic arsenic exposure is linked to increased risk of skin, lung, bladder, liver, and kidney cancer. Arsenic also causes peripheral neuropathy, cardiovascular disease, and type 2 diabetes. The latency period between exposure and cancer development can span decades, making it difficult to establish causation.
Arsenic does not require metabolic activation to damage DNA—it acts directly, making it particularly insidious during prolonged low-dose exposure.
⚙️ Synergistic Toxicity: When Metals Amplify Each Other's Effects
The simultaneous presence of multiple heavy metals in a single product creates synergistic toxicity effects. Lead and arsenic together amplify oxidative stress and DNA damage.
| Metal Combination | Interaction Mechanism | Result |
|---|---|---|
| Lead + Arsenic | Synergistic amplification of oxidative stress | Toxicity exceeds sum of individual effects |
| Mercury + Lead | Competition for detoxification systems | Reduced elimination of both metals |
| All three metals | Multiple damage pathways + depletion of antioxidant systems | Unpredictable, often critical toxicity |
Mercury and lead compete for the same detoxification systems, reducing the body's ability to eliminate both metals. Safety standards developed for individual metals do not account for these synergistic effects.
Ayurvedic products often contain not one, but two to three heavy metals simultaneously—this means actual toxicity may be several times higher than models for individual substances predict.
Data Conflicts and Zones of Uncertainty: Where Evidence Diverges
Scientific integrity requires acknowledging areas where data are incomplete or contradictory. This doesn't weaken the overall conclusion about toxicity, but shows the boundaries of our knowledge. More details in the Logical Fallacies section.
🔄 Bioavailability Variability: Not All Metal Forms Are Equally Toxic
One legitimate question concerns the bioavailability of metals in bhasma form. It's theoretically possible that some processing methods genuinely reduce the solubility and absorption of metals in the gastrointestinal tract.
However, systematic pharmacokinetic studies of bhasmas are extremely scarce. The few available data show that metals from bhasmas are still absorbed and accumulate in tissues (S002). The lack of standardization in preparation processes means bioavailability can vary significantly between manufacturers.
📉 The Problem of Missing Long-Term Cohort Studies
Most data on Ayurvedic preparation toxicity come from analyses of individual poisoning cases and cross-sectional studies of metal content (S003). Large-scale prospective cohort studies that would track users over decades and compare their health with control groups are virtually nonexistent.
The absence of long-term data doesn't mean absence of risk—it means we cannot fully assess the scale of slowly developing effects such as cancer or neurodegenerative diseases.
🌐 Geographic Variability: Quality Issues Across Countries
The quality of Ayurvedic preparations varies significantly depending on the country of manufacture and regulatory environment. Products for the domestic Indian market may differ from export products.
- Products manufactured in India often don't undergo mandatory heavy metal testing
- Export products may be subject to stricter controls, but this isn't guaranteed
- Products manufactured in Western countries are theoretically safer, but the absence of mandatory testing means consumers cannot rely on geographic origin as a guarantee
The problem is compounded by the fact that scientific databases contain a limited number of studies on quality variability between regions. This creates an information vacuum filled by marketing claims about "authenticity" and "traditionality."
⚖️ Where Interpretations of the Same Data Diverge
Even when studies show the presence of metals in preparations, Ayurveda advocates often interpret this as "contamination" rather than an intentional component. However, classical Ayurvedic texts and modern manufacturers openly identify metals as active ingredients (S008).
- Interpretation 1: "It's Contamination"
- Advocates claim metals enter preparations accidentally, from the environment or due to poor manufacturing quality. This allows separating the idea of Ayurveda from actual risks.
- Interpretation 2: "It's an Intentional Component"
- Scientific data and historical texts indicate that metals are part of traditional formulations. This means risk is built into the system itself, not a side effect of poor manufacturing.
The second interpretation aligns with toxicity mechanisms (S001) and clinical poisoning cases documented in medical literature (S006, S007).
Cognitive Anatomy of the Myth: Which Psychological Mechanisms Make People Ignore Toxicological Data
Understanding the cognitive biases that make people vulnerable to dangerous Ayurvedic products is critically important for developing effective public health strategies. More details in the Ethnic and Indigenous Identity section.
🕳️ Appeal to Nature: Why "Natural" Seems Safe
The appeal to nature (naturalistic fallacy) is a cognitive bias in which people automatically attribute positive qualities to anything perceived as "natural." Ayurvedic product marketing exploits this by positioning products as "herbal," "natural," and "ancient."
People forget that cyanide, botulinum toxin, and aflatoxins are also completely natural, yet deadly (S001). Evolutionarily, this bias may have been adaptive in an environment where unfamiliar synthetic substances genuinely represented new threats, but in the modern world it leads to irrational decisions.
The naturalness of a substance does not correlate with its safety. Toxicity is determined by molecular structure and dose, not by origin.
🎭 The Halo Effect of Tradition: Antiquity as Proof of Truth
The halo effect applied to traditional medicine causes people to transfer positive associations with "antiquity" onto specific medical practices. If a system has existed for thousands of years, it is perceived as time-tested and therefore safe.
This ignores the fact that many ancient practices (bloodletting, skull trepanation, use of mercury to treat syphilis) were dangerous and ineffective, yet persisted for centuries due to the absence of scientific methods to test them (S003).
- Antiquity ≠ effectiveness. Lack of scientific verification does not mean safety.
- The survival of a practice throughout history is explained by social and economic factors, not proven benefit.
- Modern Ayurveda often differs from the historical version, but marketing conceals this difference.
🔮 Illusion of Control Through "Natural Treatment"
Using Ayurvedic products gives people an illusion of control over their health, especially with chronic conditions where modern medicine offers only symptom management. Choosing "natural" treatment is perceived as an active, conscious decision, unlike "passively" following a doctor's recommendations.
The illusion of control is psychologically comfortable, even if it objectively increases health risks. The brain prefers the feeling of agency to danger.
📢 Confirmation Bias and Echo Chambers in Alternative Medicine Communities
People using Ayurvedic products often join like-minded communities (online forums, social media groups) where positive information about traditional medicine dominates. Confirmation bias causes them to actively seek information confirming their choice and ignore contradictory data (S006).
Reports of poisoning cases are interpreted as "rare exceptions" or "fabrications by the pharmaceutical industry." The echo chamber amplifies the effect: each new message from a like-minded person reinforces the belief.
- Echo Chamber Mechanism
- Social media algorithms show content similar to what the user has already liked. This creates an illusion of consensus: if everyone in the feed says the same thing, it must be true.
- Social Proof
- If hundreds of people in a group recommend a product, individual skepticism is suppressed. Disagreeing with the group is perceived as a social threat.
- Exit Cost
- Admitting error means losing status in the community and needing to reevaluate one's decisions. Psychologically, it's easier to stay in the group.
💰 Economic Incentives and Conflicts of Interest
Manufacturers and sellers of Ayurvedic products have a direct financial interest in their popularization. Marketing often uses emotional narratives about "ancient wisdom" and "pharmaceutical conspiracy" that resonate with distrust of institutions (S008).
This distrust is often justified (pharmaceutical companies have indeed committed ethical violations), but it is used for manipulation: if "Western medicine" is unreliable, then "natural" must be safer. Logical fallacy: criticism of one source does not automatically make an alternative source correct.
| Cognitive Bias | How It Works | Result |
|---|---|---|
| Appeal to Nature | "Natural" = safe | Ignoring toxicity of lead, mercury, arsenic in products |
| Halo Effect | Antiquity = effectiveness | Lack of critical analysis of historical practices |
| Illusion of Control | Active choice = safety | Overestimation of one's ability to assess risk |
| Confirmation Bias | Seeking information confirming belief | Ignoring scientific data on toxicity |
| Social Proof | If everyone uses it, it must be safe | Mass distribution of dangerous products |
These mechanisms work not because people are stupid, but because the brain uses heuristics (mental shortcuts) to process information under uncertainty. When data is contradictory or unavailable, the brain fills gaps with emotions, social signals, and historical narratives.
Protection from these mechanisms requires not more information, but a change in how it's processed: awareness of one's own biases, active search for contradictory data, and willingness to change one's mind when new evidence appears.
Verification Protocol: Seven Steps to Check Any "Herbal" Product Before Use
A practical checklist for minimizing risks when considering the use of Ayurvedic or other traditional products.
✅ Step 1: Verify Manufacturer Registration and Certification
Ensure the manufacturer is registered with the appropriate regulatory authority (FDA in the US, EMA in Europe, TGA in Australia, Ministry of Health in Russia). Check for GMP (Good Manufacturing Practice) certification.
Absence of registration or certification is the first signal of poor quality control. This doesn't guarantee safety, but its presence reduces the risk of heavy metals entering the product during manufacturing.
✅ Step 2: Search for Independent Laboratory Analysis
Request test results from the manufacturer for lead, mercury, arsenic, and cadmium content (S001). Verify whether the analysis was conducted by an independent laboratory, not an internal one.
If the manufacturer doesn't provide data or only references internal tests—that's a red flag. Legitimate companies publish third-party results.
✅ Step 3: Check Ingredients and Presence of Metallic Components
Review the complete ingredient list. Look for names like "rasa," "bhasma," "kalpa"—these are traditional Ayurvedic forms that often contain metals (S008).
If ingredients are listed vaguely ("herbal complex," "ancient formula") without details—demand the full list. Lack of transparency in composition correlates with hidden components.
✅ Step 4: Analyze Manufacturer Claims for Red Flags
- Promises of "miracle cures" for serious diseases (cancer, diabetes, autoimmune disorders).
- Claims that the product "detoxifies" or "removes toxins" without explaining the mechanism of action.
- References to antiquity as proof of effectiveness.
- Absence of mention of side effects or contraindications.
- Criticism of conventional medicine as a "pharmaceutical conspiracy."
✅ Step 5: Check for Clinical Research
Search for the product in databases like PubMed, Google Scholar, or scientific databases. Are there randomized controlled trials (RCTs) on humans?
Animal studies or in vitro research are not proof of safety for humans. If there are no studies or they were conducted by the manufacturer itself—this doesn't mean the product is dangerous, but it doesn't confirm its effectiveness either.
✅ Step 6: Consult with a Physician and Check for Interactions
Inform your doctor about your intention to use the product. Check for interactions with current medications through resources like Drugs.com or Medscape.
Even if the product doesn't contain toxic metals, it may enhance the effects of anticoagulants, reduce the effectiveness of antidiabetic medications, or cause allergies (S006).
✅ Step 7: Monitor After Starting Use
If you decide to use the product, conduct a blood test for heavy metals after 1–3 months. Watch for symptoms: nausea, headaches, memory problems, joint pain.
Absence of immediate symptoms doesn't mean absence of metal accumulation. Lead and arsenic accumulate in bones and organs, manifesting effects months or years later (S003).
If tests show elevated metal levels—stop taking the product immediately and consult a toxicologist or physician for chelation therapy.
